HESA Committee Report
If you have any questions or comments regarding the accessibility of this publication, please contact us at accessible@parl.gc.ca.
INTRODUCTIONIn 1921, at a medical lab at the University of Toronto, Dr. Frederick Grant Banting, his assistant Charles Best and his colleagues discovered insulin, a life-saving treatment for diabetes. This great discovery resulted in Dr. Banting and his fellow-researcher Dr. John James Rickard Macleod becoming the first Canadians to be awarded the Nobel Prize for Medicine in 1923. And yet today, almost a century later and just a few kilometres away from the Banting and Best Institute, doctors at the Sunnybrook Health Sciences Centre must maintain a supply of insulin in their offices, so they can hand it out to the five or six patients they see per month, who can no longer afford to pay for it.[1] These patients cannot afford their insulin because they have insufficient or no prescription drug coverage, a problem that is not limited to Canadians with diabetes.[2] A 2015 Angus Reid Survey found that 23% of Canadians reported that they or someone else in their household did not take their prescription medications as prescribed, if at all, because of their cost in the last 12 months.[3] Another survey conducted by the Commonwealth Fund in 2016 found that 10.2% of Canadian respondents aged 18 and older did not fill their prescriptions or skipped doses of medications due to cost in the last 12 months.[4] Furthermore, the Parliamentary Budget Officer found that 20% of Canadian households spent more than 1% of their after-tax household income on prescription drugs in 2008.[5] This trend shows no sign of abating as an increasing number of high-cost specialty drugs are being used to treat complex chronic conditions, such as cancer, rheumatoid arthritis and hepatitis C outside a hospital setting.[6] Recognizing the critical importance of this issue to Canadians, the House of Commons Standing Committee on Health (“the Committee”) agreed on 7 March 2016 to undertake a study on the development of a national pharmacare program as an insured service for Canadians under the Canada Health Act and to report the findings to the House.[7] During the course of its study, the Committee held 23 hearings, heard from 99 witnesses and received 31 written submissions and 38 reference and background documents from interested individuals and stakeholder organizations, which form the basis of the Committee’s report. In addition to his testimony, the Committee passed a motion on 29 September 2016 requesting that the Office of the Parliamentary Budget Officer (PBO) estimate the costs of providing a universal, prescription drug coverage program under the Canada Health Act.[8] On 17 October 2017, the PBO presented the findings of its report entitled Federal Cost of a National Pharmacare Program to the Committee.[9] The Committee would like to thank Jean-Denis Fréchette, Parliamentary Budget Officer and his staff for their excellent and thorough work, which has also greatly informed this report. Drawing on witness testimony and the work of the PBO, this report examines the role that the federal government could play in developing a national pharmacare program that would address the key challenges related to accessibility and affordability of prescription pharmaceuticals in Canada. The first part of the report focuses on providing background and contextual information on prescription drug coverage in Canada, such as definitions of key terms, an overview of the roles of the federal, provincial and territorial governments in prescription drug coverage in Canada and Canada’s current mix of private and public prescription drug coverage programs. It also examines current trends in prescription drug expenditures in Canada and how Canada compares internationally in these areas. The second part of the report focuses on key challenges facing Canada’s mix of public and private drug coverage and current approaches for addressing them, as well as best practices in these areas from other jurisdictions. Finally, the Committee’s report will conclude by identifying how to move forward through an examination of various policy options proposed by witnesses to expand prescription drug coverage in Canada while improving the management of drug costs. WHAT IS PHARMACARE?Unlike most member countries of the Organisation for Economic Co-operation and Development (OECD), Canada does not have a national pharmacare program – that is, a single system of public insurance coverage for prescription drugs.[10] However, there is no single model for such a program. Its design can be determined by a number of factors, including which population groups are targeted, which types of drugs are covered, and how it is financed. In terms of population coverage, a pharmacare program can be universal, meaning that it covers the whole population. Alternatively, it could target specific population groups, such as individuals with low or no incomes, individuals with specific diseases or conditions, or vulnerable population groups, such as children and seniors. A formulary is a list of drugs whose costs are covered by a drug coverage program. It also defines the conditions under which the costs of a drug may be reimbursed by the program. A drug formulary could be an open formulary which includes all drugs that have been authorized for sale, or it could be a managed formulary which covers drugs based upon certain criteria, such as covering only drugs that are considered medically necessary, meet specific population health needs, or offer value for money. A formulary covers prescription drugs and under certain circumstances, it may also cover the costs of over the counter (OTC) medications, which do not require a prescription to be purchased but may require a prescription to have their costs covered by a program. It can also include non-drug products, such as diabetic supplies. Finally, a formulary may cover brand-name drugs and generic drugs, and/or it may require that a brand-name drug be substituted with a generic drug when one is available. A brand-name drug refers to the first version of a new innovative drug to be sold and marketed, which may be patented[11] or off-patent.[12] A generic drug has the identical active ingredients as the brand-name drug but enters the market after the brand-name drug’s patent expires.[13] In terms of financing, a pharmacare program can be entirely publicly financed whereby the program covers the total cost of a drug, which is called “first-dollar” coverage. Alternatively, there can be a mix of public and private financing, whereby a portion of a drug’s cost is covered by a publicly funded insurance plan and the remainder is paid for out-of-pocket by the individual. In the design of a prescription drug coverage program, out-of-pocket payments can take a variety of forms:[14]
THE ROLES OF FEDERAL/PROVINCIAL/TERRITORIAL GOVERNMENTS IN PRESCRIPTION DRUG COVERAGE IN CANADAOne factor that will influence the design and implementation of a national pharmacare program in Canada is that jurisdiction over pharmaceuticals is shared between federal and provincial governments. Though the Constitution Act, 1867[16] does not explicitly include “health” as a legislative power assigned either to Parliament (in section 91) or to the provincial legislatures (in section 92), it does contain some powers relating directly to health and health care, including prescription pharmaceuticals. The federal government may use the criminal law power in section 91(27) of the Constitution Act, 1867, to protect the public health and safety of Canadians.[17] Consequently, the federal government, through Health Canada, is responsible for regulating the safety of pharmaceuticals, including authorizing their entry to market based upon assessments of drug safety, efficacy and quality, as well as monitoring these products once they are on the market.[18] In addition, under section 91(22) of the Constitution Act, 1867, the federal government has jurisdiction over patents. Under the Patent Act, the federal government is authorized to regulate and report on manufacturers’ prices for patented drugs through the Patented Medicine Prices Review Board (PMPRB), an arm’s-length, quasi-judicial organization that reports to Parliament through the Minister of Health. The PMPRB’s mandate is to ensure that the wholesale prices charged for patented pharmaceuticals by manufacturers are “not excessive.”[19] To carry out its mandate, the PMPRB limits increases in the price of existing patented drugs to the rate of general inflation and compares the sale price of the same drugs marketed in France, Germany, Italy, Sweden, Switzerland, the United Kingdom and the United States.[20] The jurisdiction of the PMPRB is limited to the “factory-gate” price charged by the manufacturer to wholesalers, pharmacies and hospitals, rather than the retail prices charged to consumers by wholesalers and pharmacies. However, provinces are responsible for the regulation of manufacturers’ prices for generic drugs under section 92(13) of the Constitution Act, 1867, relating to “property and civil rights in the province.” The power over property and civil rights is also the constitutional provision that grants provinces jurisdiction over health care delivery. This provision is seen as the basis for provincial authority to regulate businesses in the province, including the public and private provision of health care insurance. It also provides for the provincial regulation of health care professionals, including their prescribing and dispensing practices related to pharmaceuticals. Finally, section 92(7) grants the provinces authority to establish and regulate hospitals, as well as hospital-based health services, with the exclusion of marine hospitals. While the provinces have primary jurisdiction in health care delivery, including the prescribing and cost coverage decisions related to pharmaceuticals, the federal government has used its spending power, which is inferred from its power to raise taxes under section 91(3) of the Constitution Act, 1867, to play a role in financing in-hospital drug coverage under the Canada Health Act.[21] The Canada Health Act establishes five criteria that the provinces must observe in order to receive federal funding for their public health insurance programs through the Canada Health Transfer: public administration, comprehensiveness, universality, portability and accessibility.[22] While the Canada Health Act mandates that provinces must provide public coverage for physician services and hospital care, it requires that the cost of pharmaceuticals dispensed while in hospital be covered by publicly funded insurance plans but not the cost of prescriptions dispensed outside of hospitals.[23] The exclusion of out-of-hospital pharmaceuticals from the Canada Health Act means that provinces and territories determine the extent to which they offer public drug coverage for out-of-hospital prescription to their citizens and the overall costs of these programs. In addition to providing financing under the Canada Health Act, the federal government also provides indirect financial support to cover the costs of out-of-hospital prescription drugs by not including employer-sponsored health care benefit plans as part of an employee’s taxable income, at an estimated cost of $2.61 billion in 2016.[24] The federal government also provides the Medical Expense Tax Credit; this is a 15% non-refundable tax credit to individuals whose medical expenses amount to 3% or more of their net income, to a maximum of $2,268 for 2017.[25] There is also a supplemental refundable medical expense tax credit with a maximum amount of $1,203 in 2017. Finance Canada has estimated that these tax treatments cost the federal government $1.48 billion and $150 million respectively.[26] The “Peace, Order and Good Government” (POGG) power (also known as the residual power) referred to in section 91 of the Constitution Act, 1867, may also apply in the context of a national pharmacare program.[27] According to Professor Bruce Ryder, Associate Professor, Osgoode Hall Law School, York University, this power allows for Parliament to legislate in a matter of national concern, including health.[28] However, the subject matter of the legislation must be quite narrow and specific. Furthermore, it must also be demonstrated that provinces and territories are unable to deal with the matter effectively on their own without the federal government’s involvement. For POGG to apply in the development of a national pharmacare program, it must be demonstrated that provinces and territories are unable to provide affordable access to prescription drugs when acting independently from one another. He noted that “if you accept that argument, then I think there is a powerful basis for using the POGG national concern branch, but it would mean going out on a limb that Parliament rarely climbs out on, and it’s not completely sturdy.”[29] Finally, section 91 of the Constitution Act, 1867 also grants the federal government authority over some groups including members of the military, militia, and naval services; First Nations, Inuit and Métis; and federal inmates. Under section 95, the federal government also has jurisdiction concurrently with the provinces over immigration. Consequently, it provides or facilitates drug coverage for members of some of these groups. As an employer, the federal government also provides drug coverage to employees of the Public Service of Canada. COLLABORATION BETWEEN FEDERAL AND PROVINCIAL/TERRITORIAL GOVERNMENTS IN PRESCRIPTION DRUG COVERAGE IN CANADAGiven their shared jurisdiction over pharmaceuticals, federal, provincial and territorial governments have collaborated to manage the costs associated with their respective drug coverage programs through the establishment of three bodies: the Canadian Agency for Drugs and Technologies in Health (CADTH), the pan-Canadian Pharmaceutical Alliance (pCPA) and the Canadian Institute for Health Information (CIHI). A. Canadian Agency for Drugs and Technologies in HealthThe CADTH is an independent not-for-profit corporation established in 1989 by federal, provincial and territorial governments with the exception of Quebec. Reporting to the deputy ministers of health, the operating budget of the organization is approximately $28 million; 58% of its funding is provided by the federal government, 27% is provided by participating provinces and territories and 15% comes from other sources.[30] The CADTH is a health technology assessment agency, which provides evidence-based assessments of the clinical and cost effectiveness of drugs; diagnostics; medical, dental, and surgical devices; procedures; and programs.[31] The CADTH’s Common Drug Review Program undertakes reviews of the clinical and cost effectiveness of new and existing prescription drugs in comparison to other treatments available.[32] It then makes recommendations to federal, provincial and territorial governments as to whether or not the drug should be listed on their respective public drug plan formularies for reimbursement. The CADTH also houses the pan-Canadian oncology drug review program which reviews the clinical and cost effectiveness of oncology drugs and makes recommendations as to whether these drugs should be reimbursed by federal, provincial and territorial drug plans and/or their respective cancer agencies. However, final decisions regarding formulary listings for both cancer and non-cancer drugs rest with federal, provincial and territorial governments for their respective drug coverage programs.[33] Quebec has its own process for undertaking the clinical and cost effectiveness of both cancer and non-cancer drugs and making formulary listing decisions through the Institut national d’excellence en santé et en services sociaux (INESS).[34] B. Pan-Canadian Pharmaceutical AllianceThe pCPA was created by provincial and territorial governments in 2010.[35] The federal government joined the organization in January 2016. The pCPA undertakes drug price negotiations with drug manufacturers on behalf of federal, provincial and territorial public drug plans. The pCPA negotiates prices for brand-name drugs that have been recommended for drug formulary listing by the CADTH. Once the pCPA and drug manufacturers have reached an agreement on a price discount for a drug, a letter of intent is signed.[36] However, it still remains up to each jurisdiction whether to enter into a final product listing agreement with the manufacturer.[37] With respect to generic drugs, the pCPA has established a tiered pricing framework, which sets the price of a generic drug at 18% of the brand-name drug’s price.[38] How both the pCPA and the CADTH fit in to the overall prescription drug approval process in Canada is illustrated in figure 1. C. Canadian Institute for Health InformationThe CIHI is an independent, not-for-profit organization that collects data and provides information on Canada’s health systems.[39] It is governed by a board of directors that includes representatives of federal, provincial and territorial governments with the exception of Quebec. It is jointly funded by federal, provincial and territorial governments. The CIHI’s National Prescription Drug Utilization Information System (NPDUIS) maintains data from all provincial public drug plans except Quebec’s, as well as the federal Non-Insured Health Benefits Program for First Nations and Inuit.[40] The CIHI uses NPDUIS drug claims’ data to support public drug plans in measuring the drivers of drug use and spending, evaluate policy options, and examine potential safety concerns such as inappropriate drug use, prescription abuse and the concurrent use of multiple drugs by a patient.[41] Data from NPDUIS are also used to support the work of the PMPRB and the CADTH. Figure 1. Prescription Drug Approval and Pricing Process
OVERVIEW OF PRESCRIPTION DRUG COVERAGE IN CANADAAs out-of-hospital prescription drugs are not covered under the Canada Health Act, Canadians may be able to obtain coverage for these drugs through a mix of public and private plans. According to Ms. Abby Hoffman, Assistant Deputy Minister, Strategic Policy Branch, Department of Health, approximately 21% of Canadians obtain public drug coverage through provincial and territorial plans, which target specific groups such as seniors, social assistance recipients, individuals with certain diseases or conditions and more general plans for individuals with no other form of coverage.[42] The federal government provides drug coverage for approximately 3% of the population through plans for First Nations and Inuit, members of the Canadian Armed Forces, veterans and the RCMP, federal inmates, certain classes of refugees and federal public servants. Approximately 25.3 million Canadian residents, or 70.5% of the population obtain full or partial drug coverage through private health insurance programs, which are sponsored by employers, unions, professional associations, or are purchased individually.[43] However, Ms. Hoffman explained that, despite the provision of prescription drug coverage through a mix of public and private plans, approximately between 10% and 20% of the population do not have adequate coverage for prescription drugs.[44] In its report, Federal Costs of National Pharmacare Program, the PBO indicated that an estimated 2% of Canadians lack drug insurance coverage and 10% of Canadians who have coverage, lack the financial means to pay for their prescriptions.[45] The report further notes that the exact number of uninsured and underinsured Canadians remains “unknown, attributable to several factors including the large number of insurance providers, the various levels of benefits and coverage, and the lack of data availability.”[46] An overview of the various public and private drug coverage plans available in Canada is provided in the sections below. A. Provincial and Territorial Government Prescription Drug Coverage PlansProvincial and territorial governments offer approximately 70 different prescription drug coverage programs for their respective residents. These public drug plans fall into three main categories: catastrophic drug coverage plans, general public drug coverage plans and targeted drug coverage plans. These different types of prescription drug coverage programs are outlined in the sections below. For complete details regarding these programs, see Appendix A of this report. 1. Catastrophic Prescription Drug Coverage PlansThe phrase “catastrophic drug coverage” refers to insurance models that protect individuals from drug expenses that threaten their financial security or cause “undue financial hardship.”[47] According to the World Health Organization, “catastrophic” health expenditures are those that cannot be afforded unless a household cuts down on basic necessities such as food, clothing, or education.[48] Under a catastrophic drug coverage program, out-of-pocket expenses for prescription drugs are capped at a certain level to prevent financial hardship. This cap on out-of-pocket drug costs can be set either as a fixed dollar amount or as a percentage of personal or family income. Seven provinces (British Columbia, Saskatchewan, Manitoba, Ontario, Nova Scotia, Newfoundland and Labrador, and Prince Edward Island) offer this type of program with varying benefit payment structures (premiums, deductibles and co-payments) as well as caps on out- of-pocket payments. 2. General Public Drug Coverage PlansThe second category of provincial or territorial drug coverage plan consists of general public pharmaceutical coverage programs for individuals who may not have access to another form of drug coverage. Four provinces offer this option: Quebec, Alberta, New Brunswick and Prince Edward Island. A brief overview of each of these types of programs is provided below. i. Quebec’s Public Prescription Drug Insurance Plan[49]In 1997, Quebec established universal pharmacare by requiring residents to have drug coverage either through a private plan sponsored by their employer or professional association or through the government-run public plan. All private plans must offer the equivalent coverage of what is offered in the government public plan and cannot deny coverage or charge higher premiums because of age, sex or state of health. Both public and private plans must have caps on out-of-pocket costs. In terms of its drug formulary, Quebec’s List of Medications includes 8,000 drugs, including those for the treatment of specific conditions such as cancer, Crohn’s disease, and rheumatoid arthritis.[50] Quebec’s List of Medications is established by the Minister of Health and Social Services, in consultation with INESS. Private plans in Quebec must provide coverage for all the drugs on the List of Medications. ii. Alberta’s Non-Group Coverage Benefit ProgramThe Non-Group Coverage Benefit Program is a publicly funded supplementary health insurance program sponsored by the Government of Alberta and delivered by Alberta Blue Cross.[51] It is accessible to individuals under the age of 65 and their dependents. The drug formulary and its out-of-pocket payments structures are set by the government. iii. New Brunswick Drug Plan[52]The New Brunswick Drug Plan is for individuals who do not have existing drug coverage through their employer or another government plan, or who have other forms of drug coverage but their plan does not cover a necessary drug. iv. Prince Edward Island’s Generic Drug Program[53]Prince Edward Island’s Generic Drug Program is for individuals under the age of 65 who do not have private prescription drug coverage. The program provides coverage for generic drugs listed on the province’s drug formulary. 3. Targeted Prescription Drug Coverage Plans[54]The third category of drug coverage programs offered by some provinces and territories are programs targeted to population groups with specific needs:
B. Federal Government Prescription Drug PlansThe federal government offers six different prescription drug plans for First Nations and Inuit, members of the Canadian Armed Forces, veterans and the RCMP, federal inmates, refugees and federal public servants, which are outlined below. 1. The Department of Indigenous Services Canada’s Non-Insured Health Benefits ProgramThe Department of Indigenous Services Canada’s Non-Insured Health Benefits (NIHB) Program is a national program that provides medically necessary health benefits, to over 839,000 First Nations individuals registered under the Indian Act and Inuk recognized by one of the Inuit land claim organizations.[55] The NIHB provides drug coverage without deductibles, premiums, co-payments or user fees. Providers are encouraged to bill the program directly so that clients do not face out-of-pocket expenses. According to Mr. Sony Perron, Senior Assistant Deputy Minister, First Nations and Inuit Health Branch[56], Department of Health, approximately 514,000 NIHB clients received pharmacy benefits in 2015-2016, resulting in a utilization rate of 61% and expenditures of $427 million or $817 per capita.[57] The program’s Drug Benefit List (DBL) covers both prescription and non-prescription drugs, including vitamins, smoking cessation products, antihistamines, topical antibiotics, non-hormonal contraception methods and OTC pain medications to meet specific population health needs. According to Mr. Perron, the DBL is a managed formulary, where listing decisions are based upon clinical and cost effectiveness and safety.[58] The program typically follows the CADTH’s Common Drug Review recommendations, but also undertakes its own review through an independent Drug Therapeutics Advisory Committee that provides recommendations based upon the program’s client needs. NIHB has also signed 24 product listing agreements with drug manufacturers as a result of participation in pan-Canadian drug price negotiations through the pCPA.[59] 2. Veterans Affairs Canada’s Health Benefits ProgramThrough its health benefits program, Veterans Affairs Canada (VAC) provides prescription drug coverage to eligible veterans. Veterans’ eligibility for drug coverage depends on numerous factors such as their military service, income status or disability, or medical conditions arising from service, or whether or not they have access to other provincial/territorial drug coverage for their particular illness. According to Mr. Michel Doiron, Assistant Deputy Minister, Service Delivery Branch, Department of Veterans Affairs, the Department’s health benefits plan provided coverage to approximately 48,000 veterans in 2015-2016.[60] In that same year, expenditures amounted to approximately $92 million, or $1,916 per capita.[61] The formulary for the program is based upon recommendations made by the CADTH’s Common Drug Review, as well as its own internal formulary review committee. It covers the costs of both prescription and OTC medications. While VAC sets the policies for the program, it is administered by Medavie Blue Cross. 3. Canadian Armed Forces’ Drug Benefit ProgramAccording to Commander Sylvain Grenier, Senior Staff Officer, Pharmacy Services, Department of National Defence, the Canadian Armed Forces (CAF)’s Drug Benefit Program provides prescription drug coverage to 71,000 eligible members of the CAF.[62] In 2015-2016, the CAF spent $26.6 million for an average cost per member of $375.[63] Given that members of the CAF are excluded from the Canada Health Act because of their operational requirements, the program provides broad coverage of both prescription and OTC products. The CAF drug benefits list includes 1,605 of the over 13,000 drugs on the market in Canada. Formulary listing decisions are based upon recommendations from the CADTH’s Common Drug Review, as well as the CAF’s own Pharmacy and Therapeutics Committee which examines the drug’s applicability to the military context. Finally, approximately 90% of the prescription medications used by members of the CAF are obtained through bulk purchasing and dispensed through military pharmacies, while the remainder are purchased at private sector community pharmacies. 4. Immigration, Refugees and Citizenship Canada’s Interim Federal Health ProgramImmigration, Refugees and Citizenship Canada’s Interim Federal Health Program provides time-limited drug coverage to individuals according to their status under the Immigration and Refugee Protection Act, including protected persons, resettled refugees; refugee claimants; and certain other groups.[64] The program, which is administered by Medavie Blue Cross, provides full coverage of most prescription medications and other products listed on provincial/territorial public drug plan formularies.[65] 5. Public Service Health Care PlanThe federal government provides drug coverage to its employees, members of the RCMP, retirees and their spouses and/or partners, as well as children, through the Public Service Health Care Plan.[66] The Plan provides coverage of prescription drugs, as well as OTC medicines considered medically essential. It is provided through the Treasury Board of Canada Secretariat and is administered by Sun Life Financial. 6. Correctional Services CanadaCorrectional Services Canada provides full coverage of medications as part of the essential health care services that it provides to federal inmates.[67] C. Private Drug Coverage PlansThere are approximately 113,000 private drug coverage plans in Canada that are sponsored by employers, unions, and professional associations and/or purchased by individuals.[68] Employers that sponsor private group benefit plans for their employees include both publicly funded organizations, such as municipalities, universities, schools, hospitals, the civil service, as well as privately owned companies.[69] These plans are purchased from 132 private health insurance providers across the country.[70] According to the Canadian Life and Health Insurance Association, these plans provide drug coverage to approximately 25.3 million Canadians and 80% of the workforce.[71] For these private drug coverage plans:
OVERVIEW OF DRUG SPENDING IN CANADAA. Overall Spending on Drugs in Canada Dispensed Outside of HospitalsAccording to the CIHI, total expenditure on drugs dispensed outside of hospitals in Canada is forecast to reach $39.8 billion in 2017, or $1,086 per capita.[75] Spending on out-of-hospital prescription drugs is expected to reach $33.9 billion or $926 per capita in 2017, representing approximately 85.2% of total projected drug expenditures.[76] In its most recent report, Prescribed Drug Spending in Canada, 2017: A Focus on Public Drug Programs, the CIHI indicated that in 2017 spending on prescription drugs increased by 5.5% over the previous year.[77] Over-the-counter drugs and non-drug medical products[78] accounted for the remaining 14.8% of total drug spending in 2017, or $5.9 billion, or $160 per capita. Spending on prescription drugs by source of financing is further explained and shown in Figure 2 below. 1. Public Sector SpendingIn terms of the financing of prescription drug expenditure in Canada, $14.5 billion or 42.7% of spending was financed by the public sector in 2017, marking an increase of 5.8% over the previous year (see Figure 2).[79] Provincial and territorial programs accounted for $12.4 billion of public sector prescription spending in 2017, while federal programs for First Nations, Inuit, veterans, RCMP, federal inmates, and Canadian Armed Forces amounted to $0.76 billion. Meanwhile, public drug coverage offered through worker’s compensation programs and other social security funds amounted to $1.3 billion in 2017. In examining the breakdown of public drug program expenditure from 2015-2016, the PMPRB found that drug costs accounted for nearly three quarters (74.7%) of public drug program expenditure, followed by dispensing costs (21.8%), and reported markups (3.5%).[80] 2. Private Sector SpendingPrivate sector spending, which includes both out-of-pocket spending and spending by private insurance companies, amounted to $19.5 billion or 57.5% of total spending on out-of-hospital prescription drugs in 2017, marking an increase of 5.4% over the previous year.[81] Of this amount, $12.1 billion or 35.5% of total out-of-hospital prescription drug expenditure was financed through private health insurance, while the remaining $7.4 billion or 21.8% was paid for by individuals (see Figure 2). Though the proportion of private out-of-hospital spending on prescription drugs devoted to dispensing fees and mark-ups remains unknown, the PBO estimated that spending on dispensing fees and mark-ups for both the private and public sector amounted to $7.8 billion in 2015-2016.[82] Figure 2. Total Spending on Prescription Drugs Dispensed Outside of Hospitals by Source of Finance, Canada, 2017
Source: prepared by the Library of Parliament using data obtained from CIHI, Prescribed Drug Spending in Canada, 2017: A Focus on Drug Programs, 2017, p. 7. It is important to note that these amounts do not include public spending on drugs dispensed in hospitals, cancer agencies or other special agencies. Public spending on drugs dispensed in hospitals is covered under the Canada Health Act and are included instead in overall hospital spending. In 2015, hospital spending on drugs in Canada, excluding Quebec, amounted to $2.1 billion or 4.4% of total hospital expenditures in 2015.[83] Of this amount, $708 million was spent on cancer drugs. i. Regional Variation in Spending on Prescription Drugs Dispensed outside of HospitalsIn its report entitled, Federal Cost of a National Pharmacare Program, the PBO provided the Committee with a more detailed break-down of expenditures on prescription drugs dispensed outside of hospitals in Canada, including by region, age and income level drawing on QuintilesIMS data sets from 2015-2016 and data from Statistics Canada. Through its analysis, the PBO found that spending on prescription drugs dispensed outside of hospitals varies significantly across the country. In 2015-2016, spending on prescription drugs dispensed outside of hospitals was highest in Ontario ($11.3 billion) and lowest in Prince Edward Island ($0.1 billion).[84] Regional variations in total prescription drug spending can be attributed to a variety of factors, including specific population health needs in each province, varying demographics, prescribing habits of physicians and policy approaches to generic and patented drug pricing and negotiation.[85] There is also regional variation in the proportion of expenditure on prescription drugs dispensed outside of hospitals that was covered by public drug insurance programs in 2015-2016. The PBO found that public coverage of prescription drug expenditures was highest in Central and Western Canada ranging from 39% to 55% and lowest in Atlantic Canada, ranging from 27% to 35% (see figure 3).[86] Figure 3. Total Non-Hospital Prescription Drug Spending in CAD$ Billions, by Province and Primary Payer, 2015-2016[87]
Source: Figure prepared by Library of Parliament using PBO analysis of data from QuintilesIMS in Table 1-1 in PBO, Federal Cost of a National Pharmacare Program, p. 7. In examining average actual out-of-pocket spending on prescribed medicines and pharmaceutical products, including private insurance premiums, the PBO found that Canadian households, excluding those in the territories, spent on average $1,135 out-of-pocket in 2015. Ontario households had the lowest out-of-pocket expenditures at $823, on average, whereas Quebec households had the highest average out-of-pocket expenditures at $1,495 (see Figure 4). Figure 4. Average Out-of-Pocket Drug Costs* per Household, by Province, 2015
Source: prepared by the Library of Parliament using data from Figure 1-4 in PBO, Federal Cost of a National Pharmacare Program, p. 22. *This indicator does not include out-of-pocket spending on over-the-counter drugs. ii. Variation in Prescription Drug Spending by Age GroupAccording to the PBO, total spending on prescription drugs dispensed outside of hospitals varies by age in Canada, with seniors aged 65 and older accounting for 37% or $10.6 billion of total non-hospital drug spending in Canada in 2015-2016, whereas working-aged Canadians between 15 and 64 years of age accounted for 60% or $17.2 billion of total non-hospital drug spending that same year. Children and youth aged 0 to 14 accounted for the remaining $0.8 billion, or 3% (see Figure 5). Figure 5. Prescription Drug Spending by Age Group in Canada, 2015
Source: prepared by the Library of Parliament using data from Table 1-2 in PBO, “Federal Cost of a National Pharmacare Program, p. 8. According to the PBO, the share of drug spending paid for by public and private insurance, as well as by individuals also varies by age in Canada. This variation reflects the targeted coverage on the basis of age, particularly for seniors aged 65 and older, of many provincial and territorial public drug coverage programs. Approximately, 70% or more of drug spending for seniors aged 65 years and older was publicly financed in 2015-2016. In addition, seniors also spent less out-of-pocket as a portion of their total drug spending, ranging from 11% to 13% of total spending, reflecting the universal coverage generally offered to seniors through provincial and territorial programs and low co-payments, premiums and/or deductibles. However, seniors’ actual out-of-pocket costs at $646 per capita in 2015, were on average almost four times higher than those of individuals under 30 years of age because of seniors’ greater health needs (see Figure 6). In comparison, working-aged Canadians aged 15 years or older had less of their prescription drug costs covered by public plans in comparison to seniors. Approximately, 19% to 30% of prescription drug costs for working-aged Canadians aged 15 or older were publicly covered between 2015 and 2016. Meanwhile, approximately 50% of their drug costs were covered by private insurance, reflecting the fact that this age group tends to have coverage through their employers. In addition, working-aged Canadians must also pay a greater proportion of their drug costs out-of-pocket in comparison to seniors, ranging from 18% to 20% of their total non-hospital prescription drug costs. Out-of-pocket costs for working-aged Canadians were highest for individuals over the age of 55 in 2015 (see Figure 6). Figure 6. Average Reported Out-of-pocket Spending on Prescribed Medicines and Pharmaceutical Products, by Age Group, 2015
Source: PBO analysis of Statistic Canada’s Survey of Household Spending in PBO, Federal Cost of a National Pharmacare Program, p. 22. Notes: These out-of-pocket costs do not include premiums paid to private insurance companies, which is why the national average for out-of-pocket costs differs from what is provided in Figure 3. ** Reference Person is the household member that is mainly responsible for the financial maintenance of the household. iii. Financial Burden of Prescription Drug Spending on Households by IncomeThe PBO also examined spending on prescription drugs dispensed outside of hospitals by income level based upon Statistics Canada’s Survey of Household Spending, 2015. It found that there were no significant differences in out-of-pocket spending among all five income level groups or quintiles examined. The national average for out-of-pocket spending in 2015 was $417 per household and ranged from $357 to $459. However, for the poorest households in Canada, these out-of-pocket costs represented a greater financial burden as they accounted for over 1% of their before-tax income, a financial burden that is four times greater than that facing the richest Canadian households, whose out-of-pocket prescription costs represented only 0.24% of their before-tax income (see Figure 7). Figure 7. Average Portion of Household Spending for Prescribed Medicines and Pharmaceutical Products, by Before-Tax Household Income Quintile, 2015
Source: Prepared by Library of Parliament based upon PBO analysis of Statistics Canada Data found in Figure1-6 in PBO, Federal Cost of a National Pharmacare Program, p. 23. B. Current Trends in Prescription Drug Spending in CanadaThe Committee heard from representatives from the CIHI and the PMPRB that prescription drug expenditures in Canada are influenced by various factors, including changes in the price of drugs, demographic effects, the volume of prescriptions and the types of drugs prescribed. Though Canada has reduced spending on prescription pharmaceuticals by obtaining price reductions for both generic and patented medicines through the pan-Canadian Pharmaceutical Alliance, these reductions in price have been offset by increased spending on high-cost specialty drugs, such as biologics, oncology drugs and drugs for rare diseases, which are placing an increased strain on the budgets of both public and private plans.[88] According to Ms. Tanya Potashnik, Director, Policy and Economic Analysis Branch, the PMPRB, Canadian spending on biologics and oncology drugs grew by double digits, and spending on new drugs alone increased tenfold in 2014, with new curative drugs for hepatitis C accounting for a significant proportion of these rising costs.[89] Dr. Christopher McCabe, Capital Health Research Chair, Faculty of Medicine and Dentistry, University of Alberta explained that though the individual budgetary impact of a drug for rare diseases[90] is small at around $100,000 per patient per year, their overall budgetary impact is greater when access to the drug is expanded to all eligible patients. Dr. McCabe explained that the impact of drugs for rare diseases on the budgets of public and private drug plans is expected to increase “with the literally hundreds of orphan drugs coming down the line.”[91] Other factors, such as changing demographics, also play a significant role in rising drug expenditures among public drug plans (see Table 1 for further details). Table 1. Main Cost Drivers of Public Drug Plans, 2015-2016
Source: prepared by the Library of Parliament using data from the PMPRB, “Annual Report, 2016,” 2017, p. 50. Finally, the development of high cost treatments for very specific conditions (costing more than $10,000 per individual) means that an increasing portion of private and public drug plan spending is concentrated on a few individuals. For public plans, 2% of beneficiaries for whom drug costs amounted to $10,000 per year accounted for one-third of spending in 2016.[92]These beneficiaries were receiving new high cost treatments primarily for chronic conditions, including hepatitis C, rheumatoid arthritis and Crohn’s disease.[93] Similarly, 14% of members of private plans represented 72% of total expenditure by private insurance companies on claims, while members with claims exceeding $10,000 per year accounted for 28.8% of total spending in 2016.[94] According to Express Scripts Canada, members of private plans with claims exceeding $10,000 are individuals managing multiple chronic conditions, which may also require high cost treatments for these conditions.[95] HOW DOES CANADA COMPARE INTERNATIONALLY?The Committee heard from representatives from both the CIHI and the PMPRB that Canada’s spending on prescription drugs is significantly higher than that in other OECD countries.[96] In 2015, Canada’s per capita drug expenditure ranked third highest among 29 OECD countries, behind the United States and Switzerland. At $1,012, Canada’s per-capita expenditure on drugs, including pharmaceutical goods and other medical non-durables, was approximately 35% higher than the OECD average (see figure 8). Figure 8. Total expenditure on drugs per capita, Canadian dollar, purchasing power parity, 29 selected OECD countries, 2015
Source: CIHI, “Information Sheet: Drug Spending at a Glance,” 2017. According to the PMPRB, Canadian patented drug prices were also third highest among OECD countries in 2016, behind the United States and Switzerland (see Figure 9). Ms. Potashnik further explained to the Committee that despite Canada’s relatively high patented drug prices, research and development (R&D) activities by pharmaceutical companies as a percentage of sales remain at 5%, which is significantly lower than the average R&D to sales ratio among international comparator countries at 20%.[97] Meanwhile, generic drug prices in Canada declined substantially between 2010 and 2015, but remain 9% higher than the OECD average.[98] Figure 9. Average Foreign-to-Canadian Price Ratios[99], Patented Drugs, OECD, 2016
Source: PMPRB, “Annual Report, 2016,” 2017, p. 34 Unlike most countries with universal health care systems, Canada does not have a universal public system of prescription drug insurance. In terms of public coverage of prescription drug costs, Canada ranked 26th of 28 OECD countries behind the United States, covering only 36.3% of the costs of drugs, including pharmaceutical goods and other medical non-durables in 2015.[100] According to the 2016 Commonwealth Fund International Health Policy Survey, 10.2% of Canadian respondents did not take their medications because of their cost, a rate that was second highest among countries surveyed (see Figure 10). In his appearance before the Committee, Mr. Glenn Monteith, Vice-President, Innovation and Health Sustainability, Innovative Medicines Canada also pointed out that public plan coverage of new innovative medicines remains below that of other countries.[101] Based upon a study conducted by Innovative Medicines Canada, of the 121 new medicines approved for sale in Canada between 2010 and 2014, only 37% received public reimbursement as of 31 December 2015, which ranked Canada 18th of the 20 countries in the study.[102] Figure 10. Percentage of adults not filling a prescription or skipping doses because of cost, by country, 2016
Source: CIHI data tables: How Canada compares: results from the Commonwealth Fund’s 2016 International Health Policy Survey of Adults in 11 countries, submitted by Dr. Marc-André Gagnon, Associate Professor, School of Public Policy and Administration, Carleton University. Witnesses appearing before the Committee explained that Canada’s mix of private and public prescription drug coverage is facing significant challenges and is in need of serious reform. In particular, witnesses identified the need to address gaps in prescription drug coverage; the variation among drug formularies across the country; the rising costs of prescription pharmaceuticals; and the burden that private drug plans pose on employers and employees. In addition, witnesses highlighted overprescribing by health care providers and limited data and information systems as critical issues to be addressed as part of any drug coverage reform efforts. An overview of these issues is provided in the sections below. A. The Prescription Drug Coverage GapAccording to witnesses, one of the main issues facing Canada’s provision of prescription drug coverage is that many Canadians do not have any form of drug coverage. According to Ms. Abby Hoffman, Canada’s mix of private and public drug coverage “leaves about 10% of Canadians without any practical form of ongoing coverage.”[103] These individuals must therefore pay the entire costs of their medications themselves.[104]
The Committee heard that individuals who have relatively low incomes or who are in part-time or precarious work arrangements are least likely to have access to drug coverage through their employers. Mr. Victor Elkins, Regional Vice-President for British Columbia, Canadian Union of Public Employees (CUPE) explained to the Committee that only 32% of individuals earning between $10,000 and $20,000 receive health benefits from their employer.[105] Meanwhile, only 27% of part-time employees receive health benefits from their employer in comparison to 73% of full-time employees.[106] Dr. Danielle Martin, Vice-President, Medical Affairs & Health System Solutions, Women’s College Hospital, explained the prescription drug coverage gap is not limited to individuals with low incomes, but also includes individuals with higher incomes who are self-employed or who work for small businesses that do not provide health benefits.[107] Finally, a written submission from the Wellesley Institute pointed out that there are gender differences in terms of access to employer-based drug coverage: 61% of female employees have access to employer-based drug coverage in comparison to 67% of male employees.[108] It explained that these gender differences may result from the greater likelihood of women occupying part-time positions. Finally, women without coverage may also receive benefits through their spouse’s employer-provided plan. However, this can place them in a vulnerable position if their relationship status changes or if their spouse’s employment situation changes.[109]
In addition to individuals without any prescription drug coverage, the Committee heard that “a further 10% of Canadians could generally be considered to be underinsured. These are people who have very high drug costs that exceed the limits of their drug plan, and that leaves them with very significant out-of-pocket costs.”[110] The Committee heard that individuals with chronic conditions or illnesses are more likely to be underinsured, often paying higher premiums, reaching monthly or annual coverage maximums, or being denied coverage altogether.[111] According to witnesses, Canadians who either have insufficient or no prescription drug coverage have high out-of-pocket drug costs and may skip taking their medications as a result, leading to poor health outcomes. In its analysis, the PBO found that approximately 20% of households in Canada in 2008 spent more than 1% of their after-tax income on prescription drugs and a further 3% of households spent more than 5% of their after-tax income on prescription drugs.[112] In Saskatchewan, Manitoba, Quebec and the Atlantic provinces, more than 20% of households spent 1% of their after-tax income on prescription drugs in 2008.[113] Ms. Shachi Kurl, Executive Director, Angus Reid Institute, further explained that these out-of-pocket costs resulted in 23% of Canadians reporting, in 2015, that they or someone else in their household did not take their prescription medications because of cost.[114] According to Dr. Jan Hux, Chief Science Officer, Canadian Diabetes Association, individuals with chronic diseases are more likely to be affected by out-of-pocket costs with one study showing that 23% of individuals with chronic disease skip medication because of cost compared to 10% in the overall population.[115] She explained that if individuals with diabetes skip their medications, it could increase their risk of long-term complications from the disease, such as blindness, amputation and heart disease. The Health Charities Coalition of Canada also shared with the Committee stories of the patients whose health conditions deteriorated because they could not afford their medications:
A doctor continues to have repeat visits from a patient who has chronic obstructive pulmonary disease. The patient is experiencing severe exacerbations that are bringing him back into the clinic repeatedly, and occasionally into the emergency room. The doctor is concerned that the prescribed treatment is not working, until one day his patient confesses that he's only been using his inhaler once a day rather than twice a day, as prescribed. Why? Because he can't afford to renew his prescription. He thought he would reduce the number of times he took it per day and make it last a little bit longer.… These are the stories we hear every single day. So what can we do?[116] The Committee heard from witnesses that out-of-pocket costs for prescription drugs lead not only to poor health outcomes for Canadians, but also result in significant costs to health care systems in Canada. Dr. Monika Dutt, Chair, Canadian Doctors for Medicare estimated that individuals not taking their medications because they cannot afford them, costs health care systems in Canada between $7 billion and $9 billion per year.[117] Dr. Dutt explained that a study in the United States demonstrated that providing medications free of charge to prevent heart attacks reduced vascular events by 11% and resulted in a reduction of health care costs of US$5,700 per patient on average.[118] She also pointed out that the control group in this study had drug coverage but had to make small co-payments of $10, illustrating that even small out-of-pocket payments could result in individuals not taking their medications. She explained that this study was replicated in Ontario with similar results.
The Committee heard that, in recent years, provincial and territorial governments have attempted to address gaps in prescription drug coverage by offering catastrophic drug coverage programs to individuals who face high drug costs relative to their income.[119] However, the eligibility requirements for these programs, which range from having out-of-pocket drug costs between 1.25% to 12% of income, still mean that individuals could be paying $1,000 out-of-pocket before a public plan begins covering costs, leaving individuals without any practical form of on-going drug coverage.[120]
For these reasons, there was unanimous agreement among stakeholders appearing before the Committee, including patient groups, health care providers, the private insurance industry, innovative drug manufacturers, unions, employers and academics that the gap in prescription drug coverage in Canada and the inequity that it creates among Canadians needs to be addressed. While witnesses differed on whether gaps in coverage should be addressed through the expansion of current programs, or the creation of a targeted program, the vast majority agreed that they should be addressed through the development of a national universal pharmacare program. The various proposed options will be discussed in greater detail in subsequent sections of the report. B. Variation in Drug Formularies Among Prescription Drug Coverage PlansIn addition to the inequities created by gaps in prescription drug coverage, the Committee heard that Canadians also face inequities because of variations in the drug formularies among different prescription drug coverage plans. The Committee heard from witnesses that though there is some commonality among the drug formularies of provincial and territorial drug coverage plans, they vary significantly in their listing decisions with respect to more expensive specialty drugs such as biologics, cancer drugs and drugs for rare diseases.[121] For example, according to Ms. Natasha Mistry, Director, Canadian Association of Retired Persons, a report by the Gastrointestinal Society found that access to biologic treatments for individuals with inflammatory bowel disease varies across provinces because of different access criteria on provincial formularies for these drugs.[122] The Committee heard that the variation in drug listing decisions among provincial and territorial programs also means that federal drug coverage programs face challenges ensuring that their drug formularies are in line with those in the provinces and territories.[123] In a background document submitted to the Committee, the Assembly of First Nations (AFN) pointed out that variation in drug listing decisions and access criteria among federal, provincial and territorial drug coverage programs create inequity and access barriers for First Nation clients of the federal NIHB program.[124] The AFN explained, for example, that Manitoba’s drug formulary covers Apretitant, a drug taken to prevent nausea and vomiting prior to chemotherapy. However, this same drug is listed as an exceptional drug on the NIHB formulary, meaning that it requires special approval, creating delays in access to care for First Nations clients.[125] Another problem raised by the Canadian Medical Association is variation between in-hospital and out-of-hospital drug formularies within provinces and territories, particularly in relation to new cancer drugs that are often prescribed and taken in hospital but can now be dispensed and taken in an out-of-hospital setting.[126] The Committee heard that while patients obtain full coverage for these drugs while they are in hospital, they may not be covered by their provincial drug plans, once they are discharged from hospital. To address this issue, Dr. Brian O’Rourke, President and Chief Executive Officer, the CADTH, explained to the Committee that the CADTH is negotiating with the provinces and territories to expand its mandate to examine both in-hospital and out-of-hospital drugs to ensure that formularies will be consistent across hospital and public drug plan sectors.[127] He further noted that additional resources would be necessary to expand the organization’s mandate in this area. Finally, the Committee heard from witnesses that there is significant variation between the drug formularies of public and private plans, as private drug plans often have open formularies with few restrictions on the number and types of drugs prescribed. The Committee heard that under these plans, patients have greater access to medications and on a more timely basis than under public plans. For example, Mr. Monteith explained that a study showed that of the 464 new drugs approved by Health Canada from 2004 to 2013, 89% were covered by at least one private plan, while only 50% were covered by at least one public plan as of 31 January 2015.[128] In terms of timeliness, Mr. Frank Swedlove, President and Chief Executive Officer, Canadian Life and Health Insurance Association, indicated that a 2012 study by the Canadian Health Policy Institute, a private think tank,[129] found that public plans took 312 days on average to make a drug listing decision, whereas private plans only took 143 days.[130] Witnesses appearing before the Committee therefore called for greater harmonization among the drug formularies of prescription drug coverage plans in Canada, which they believed could be achieved through the creation of a common national formulary. In his testimony, Mr. Swedlove further elaborated that the private drug insurance industry supports the creation of a common national minimum formulary, which would ensure a baseline of coverage for all Canadians.[131] He further noted that there needs to be a common approach across public and private plans to provide reimbursement for drugs for rare diseases because they target small population groups, but have very high costs. Patient groups are also supportive of a common national formulary. However, they emphasized that the national formulary should not be too restrictive and allow for patient choice because individuals respond to medications differently, particularly in the case of the newer more expensive biologic drugs.[132] Mr. Jim Keon, President, Canadian Generic Pharmaceutical Association, indicated that drug manufacturers also support greater harmonization among drug formularies of public and private plans, as it would reduce the duplication caused by the varied listing processes employed by each province and territory, which increase administrative costs for both manufacturers and drug plans.[133] Finally, Dr. O’Rourke indicated that his organization would be well placed to contribute to the development of a national formulary.[134]
C. Managing the Rising Costs of Prescription DrugsThe Committee heard that public and private prescription drug coverage plans in Canada are facing difficulties managing the rising costs of prescription drugs. Though Canada has mechanisms in place such as the CADTH, the PMPRB and the pCPA to manage the costs of prescription drugs, the Committee heard from witnesses that Canadians pay more for prescription drugs and face higher drug prices than residents of most other OECD countries. As noted earlier in the report, Canada’s spending on pharmaceuticals and patented drug prices were between second and fourth highest in the world in 2015 and 2016. Witnesses appearing before the Committee identified ways in which Canada’s current approaches to managing prescription drug costs could be improved to achieve greater cost savings and improve quality of care. Improved approaches would also allow prescription drug coverage plans in Canada to respond more effectively to the rising costs of new specialty drugs, such as biologics, cancer drugs and drugs for rare diseases. 1. The pan-Canadian Pharmaceutical AllianceThe Committee heard from witnesses that the pan-Canadian Pharmaceutical Alliance (pCPA) had been successful in achieving significant savings through drug price negotiations with both patented and generic drug manufacturers. According to Mr. W. Neil Palmer, President and Principal Consultant, PDCI Market Access, the pCPA had negotiated price reductions for over 100 patented products on behalf of public drug coverage programs, resulting in a cost saving of half a billion dollars annually.[135] In addition, the Committee heard that the pCPA had established a national generic tiered pricing framework that reduced the prices of 18 of the top-selling, high-volume generic prescriptions to 18% of the brand-name drugs’ price, resulting in a savings of $1.6 billion over the life of the agreement with the Canadian Generic Pharmaceutical Association.[136] The Committee heard that the pCPA’s price reductions on generic drugs are accessible to public and private drug coverage plans in Canada. Despite its success, Dr. Doug Coyle, Professor and Interim Director, University of Ottawa, School of Epidemiology, Public Health and Preventative Medicine, explained to the Committee that the pCPA could achieve greater reductions in drug prices, if it had an established transparent framework for its price negotiations, including specific criteria for determining whether the price of a drug established by manufacturers reflects value for money in terms of the health benefits it provides.[137]
He explained that the pCPA appears to accept the price reductions offered to it by manufacturers rather than negotiate further price reductions based upon scientific evidence regarding the benefits of the drugs. The pCPA also negotiate drug prices with little transparency or public oversight so the public is unable to determine whether the pCPA is actually obtaining value for money. For example, Dr. Coyle pointed to the pCPA’s negotiations for the drug Soliris, which costs $500,000 per patient per year. Though the pCPA received a deduction in price for the drug, independent analysis showed that the drug would only be worth funding if a price reduction of 98.5% was achieved.[138] While witnesses appearing before the Committee expressed differing views on the benefits and drawbacks of the pCPA’s confidential drug price negotiation process, they did agree that there needed to be a “set of principles to make sure that we are making the best possible decisions about which drugs get onto public funded formularies and which don’t. Those principles should be transparent. They should be vetted with decision-makers and, most importantly, vetted with the public and with patients so that we're not disadvantaging people with rare diseases, or disadvantaging people who have certain conditions versus other conditions.”[139] More significantly, the Committee heard that Canada could obtain further reductions in drug prices, if the pCPA negotiated drug prices on behalf of the entire Canadian drug market, rather than for public drug plans only. The Committee heard that the main drawback of the pCPA is that the savings it obtains are only for individuals who have coverage through public plans, not those who have coverage through private plans or individuals with no insurance.[140] Furthermore, as public plans represent only approximately 40% of the Canadian drug market, the pCPA lacks the bargaining power it could have if it were to represent the entire Canadian drug market:
If the drug plan [manager] is sitting across the table from manufacturers and saying, “It’s our price or you won’t be subsidized for Canadians across the country,” that’s a strong bargaining position. I think you can have rules surrounding the way these negotiations are conducted that would make them acceptable, but if you’re going to have them, you should make sure all Canadians are benefitting from them.[141] Dr. Steven Morgan, Professor, School of Population and Public Health, University of British Columbia explained that if Canada consolidated its purchasing power through the creation of a single payer universal drug coverage system covering the entire population, Canada could save upwards of $7 billion per year based upon conservative estimates.[142] Alternatively, Mr. Frank Swedlove explained that the pCPA could also expand its purchasing power by allowing private insurers to join the organization to obtain the same lower drug prices for private drug plans.[143] Other witnesses indicated that the inclusion of private plans in the pCPA may not be acceptable as they operate on a for-profit basis rather than on the interests of the public health care system as a whole.[144] Moreover, private plans are also able to obtain price reductions through their own price listing negotiations with manufacturers.[145] 2. Market Practices in the Pharmaceutical SectorThe Committee also heard that the market practices of the pharmaceutical sector should be investigated as they may also contribute to high drug prices in Canada. In particular, witnesses explained that the Competition Bureau should investigate all mergers and acquisitions among pharmaceutical companies and examine their impact on the market place and drug prices.[146] The PBO’s report further explained how generic drug companies compete to have their products stocked in pharmacies by offering confidential rebates to pharmacies, which provide pharmacies with a profit as pharmacies are reimbursed by drug coverage plans at a higher price.[147] Though these practices have been curtailed in recent years, Professor Amir Attaran, Professor, Faculty of Law, University of Ottawa explained that they continue to be reinforced by the generic tiered pricing strategy of the pCPA which sets a percentage price ceiling for reimbursement of generic drugs rather than forcing manufacturers to compete and offer lower prices.[148] This profit margin allows manufacturers to offer rebates to pharmacies, but these costs are then passed on by pharmacies to consumers and drug plans.
As a part of its deliberations, the Committee also heard how access to over-the-counter medicines, which do not require a prescription, can improve the overall access to affordable medicines for Canadians, while reducing overall healthcare costs.[149] To illustrate the need for flexible solutions tailored to meet different needs, Consumer Health Products Canada (CHPC), the organization representing the makers of non-prescription drugs and natural health products, described the important role that these medicines play in self-care, an increasingly important part of the healthcare system. This issue is relevant to improving access to affordable medicines in two ways. First, OTC medications are generally significantly less expensive than prescription drugs, makes them a more affordable option for Canadians who currently have no drug coverage, or poor drug coverage. Second, should a universal pharmacare program be created, the availability of more self-care options would reduce the demand for prescribed medicines, reducing the costs of a universal pharmacare program. According to CHPC, improved access to OTC medications and their associate cost savings can be achieved by facilitating the transition of drugs from their status as prescription drugs to being rescheduled as OTC drugs, when it is safe to do so.[150] While Health Canada is responsible for approving a drug’s change in status from prescription to non-prescription medicine under the Food and Drugs Act, the manufacturers of OTC medicines in Canada must go through an additional approval process at the provincial level, which determines the conditions of sale of the product in pharmacies and/or other locations, a process that can take up to two years. The organization therefore recommended that the federal government play a leadership role in engaging the provinces in a dialogue to identify ways of integrating the rescheduling of prescription drugs to over-the-counter status and their conditions of sale at the provincial and territorial level.[151] 3. Patented Medicine Prices Review BoardThe Committee heard from Ms. Tanya Potashnik that the board is a consumer protection agency, whose mandate is to ensure that the prices of patented medicines in Canada are not excessive.[152] However, she explained that the agency’s approach to regulating prices is no longer effective in the current drug market, which is seeing increased development of high-cost specialty drugs, including biologics, drugs for rare diseases and oncology drugs. These drugs often enter the global market to address unmet health needs where no other therapeutic options had been available to date, resulting in historically high price tags of between $500,000 and $700,000 per year per patient. While the agency normally relies on international drug price referencing to determine whether prices in Canada are considered excessive, she explained that this method no longer works as other jurisdictions are obtaining confidential rebates on drug prices such that the actual prices of these drugs in other jurisdictions remain unknown. Consequently, she explained that the PMPRB is undertaking consultations to update the organization’s legislative mandate to respond to these pressures. Witnesses appearing before the Committee agreed that the PMPRB’s mandate needs to be reformed and the organization needs to find a new approach towards capping drug prices. The Canadian Life and Health Insurance Association suggested that the new mandate of the organization should be to establish price ceilings that are the lowest possible rather than simply “not excessive.”[153] Mr. Ake Blomqvist, Health Policy Scholar, C.D Howe Institute, recommended that the PMPRB adopt value-based pricing, whereby the price of a drug is based upon evidence of improvement that the drug provides in terms of quality of life and the additional number of years lived.[154] However, the Committee also heard that this approach could discriminate against seniors because of its focus on improvement in length of life.[155] Another possible approach is the use of reference-based pricing, which has been adopted by British Columbia’s Pharmacare Program. This approach involves only reimbursing the lowest priced drug in a class of drugs that have the same therapeutic effects. According to Dr. Thomas Perry, Chair, Education Working Group, University of British Columbia Therapeutics Initiative, a 1997 study examining the use of reference-based pricing for ACE-inhibitor drugs found that it resulted in a 19% reduction in costs without any harm to health.[156] 4. Generic SubstitutionThe Committee heard that one of the cost drivers of pharmaceutical expenditures is that Canada does not make sufficient use of generic drugs. Mr. Jim Keon explained that generic drugs play a critical role in reducing drug costs by providing an equivalent, safe alternative to patented drugs at a lower price.[157] He explained that a PMPRB 2015 report entitled Compass Rx found that the use of generic drugs had saved Canadian governments, employers, and patients nearly $15 billion in the previous year. It further estimated that for every 1% increase in the use of generic drugs, Canadians would save an extra $434 million.[158] However, he noted only 69% of prescriptions in Canada are filled with generic drugs compared to 89% in the United States. According to Mr. Keon, one of the reasons for the lower rates of generic drug use in Canada is while public drug plans require that a brand-name drug be substituted with a generic drug when available, private plans do not.[159] In its report, the PBO estimated that approximately 20% of private plans do not require generic substitution for brand-name drugs available on their formularies.[160] The PBO further found that if private plans were to adopt the generic substitution rates of public plans, total prescription drug expenditures in Canada would decrease by $532.8 million.[161] The PBO report also explained that some branded prescription drug manufacturers offer co-payment cards to patients, which pay the difference in price between a brand-name and a generic drug in situations where a patient’s drug plan reimburses the equivalent of the cost of the generic substitute only.[162] Mr. Keon also explained that there is some resistance among patients and providers to adopting subsequent-entry-biologics, which are generic forms of biologic drugs, because there are fears that they are not equivalent to the originals. He suggested that these concerns possibly have been “fomented by some of the originator companies that have been selling these products for more than 20 years.”[163] 5. Canadian Agency for Drugs and Technologies in HealthWitnesses explained to the Committee that health technology assessments (HTA) are a critical component of managing the costs of prescription drug programs while ensuring quality of care. Dr. Perry explained that HTAs are used to examine evidence about how effective drugs are in comparison to other drugs or treatments in order to determine which drugs should be listed on the drug formularies of prescription drug coverage plans. HTAs achieve cost-savings for pharmacare programs by ensuring that they cover only the drugs that are most effective relative to their cost and that improve quality and safety of care. Dr. Perry provided the Committee with an example of how HTA works in practice to save money and improve patient care. In 1999, the Therapeutics Initiative undertook a review of new nonsteroidal anti-inflammatory drugs (NSAIDs) that came on the market, such as Celebrex and Vioxx. It found that these new drugs were not safer or were, in some cases, more dangerous than existing NSAIDs. As a result, B.C’s Pharmacare program did not agree to pay for the new drugs, thus reducing their overall consumption in the province in comparison to other jurisdictions which did provide coverage of these drugs. As a result, the province had fewer patients with negative side-effects resulting from the drugs, such as hospitalizations for gastrointestinal bleeds. While witnesses noted that the CADTH has been effective in conducting such cost-effectiveness research for traditional pharmaceuticals and providing drug formulary listing recommendations for federal, provincial and territorial plans, the Committee heard that it lacked the capacity to meet current and future demands. Dr. O’Rourke explained that the organization is unable to keep up with current requests from public plans to do more intensive complex therapeutic analysis of newer more expensive drugs and drugs that have the potential for abuse.[164] He further explained that the cancer agencies are also requesting reviews of cancer drugs, but the organization is not sufficiently resourced to undertake these reviews. Other witnesses suggested that the CADTH needs to develop new approaches to HTAs and drug listing recommendations to address the challenges posed by new high-cost speciality drugs and drugs for rare diseases. Professor Matthew Herder, Associate Professor, Faculties of Medicine and Law, Health Law Institute, Dalhousie University, explained that speciality drugs and drugs for rare diseases are unique because they are developed through the use of genomics to provide targeted treatments to subsets of the disease populations based upon the particular genetic profile of the disease or individual.[165] However, this targeted approach to drug development means that these new drugs are used only in small patient population groups. In addition, evidence supporting their use is limited because it does not include the large-scale clinical trials among the broader population to evaluate their safety and effectiveness, as is the case with more traditional medicines. As specialty drugs and drugs for rare diseases are used only in very small population groups, prices charged by drug manufacturers for them are relatively high, ranging from $200,000-300,000 per year per patient.[166] It is difficult for organizations such as the CADTH to evaluate and make recommendations regarding their cost-effectiveness relative to other drugs and treatments, because often there is limited evidence supporting their use, but there are often no other treatments available either, creating additional pressure to provide reimbursement for these drugs. According to Professor Herder, this situation could be addressed by building capacity within the CADTH to support provincial, territorial and federal governments in the negotiation of “performance-based risk-sharing agreements,” or “managed entry agreements” with drug manufacturers.[167] Under these types of agreements, the decision to reimburse a drug is based upon an agreement that there will be additional data collected and analyzed regarding the benefits of the drug and the price of the drug will either be increased or lowered depending upon health outcomes observed. Dr. Christopher McCabe further elaborated that these drug reimbursement agreements could also be tied to specialized health networks across the country providing care to patients receiving treatments from these drugs.[168] Finally, the Committee heard that the CADTH is poorly integrated into drug safety evaluation and reimbursement processes. Though the CADTH makes drug formulary listing recommendations, witnesses suggested that it “lacks teeth” as an organization because the federal and provincial/territorial governments decide whether they follow the CADTH’s recommendations, leading to variations in drug listing decisions across the country.[169] Furthermore, though the CADTH provides support to the pCPA for drug price negotiations, making drug price recommendations is not part of its mandate. In addition, it often lacks accurate pricing information from the provinces when undertaking its reviews.[170] Witnesses also explained that there is a need for greater information sharing between the CADTH and Health Canada so that post-market data on adverse drug reactions can be incorporated into the CADTH’s therapeutic evaluations.[171] 6. Canadian Institute for Health InformationAccording to Mr. Brent Diverty, Vice-President of Programs, CIHI, a critical component of managing prescription drug spending is having accurate data and information systems to track drug utilization rates to understand cost drivers and forecast future trends, as well as examine safety concerns such as inappropriate drug use, prescription drug abuse and the concurrent use of multiple medications by one patient.[172] However, he explained that Canada lacks comprehensive data on drugs used by all Canadians, including individuals with private insurance or without any drug coverage. The organization also lacks comprehensive drug data from all provinces and territories; it has complete data sets from British Columbia, Saskatchewan and Manitoba only. Mr. Diverty said that greater collaboration among governments, health system stakeholders, and the private sector is needed in order to expedite the creation of a complete data set. According to Dr. Robyn Tamblyn, Professor, Department of Medicine and Department of Epidemiology, Biostatistics and Occupational Health, McGill University, one possible way of addressing this issue is through the creation of a national real-time electronic adverse drug reaction reporting system, which could integrate the CIHI’s data holdings with the national electronic prescription monitoring platform, which is currently being developed by Canada Health Infoway as part of the Electronic Health Record systems.[173]
D. The Increasing Burden of Private Drug Plans on Employers and EmployeesThe Committee heard from witnesses that the rising costs of prescription drugs are also placing a strain on employers who sponsor private drug insurances plans for their employees as part of their overall compensation package. Some witnesses told the Committee that private plans are passing off the costs of prescription pharmaceuticals onto employers and employees in the form of rising premiums, deductibles and co-payments, rather than improving the management of these programs.[174] For example, witnesses pointed to a study by Express Scripts Canada that found that private plans provided $5.1 billion in reimbursements for drugs that offered no therapeutic benefit.[175] As employers themselves are not in the best position to rein in these costs, the Committee heard that they are instead beginning to reduce the drug benefits offered to employees by providing flexible plans, which require employees to determine how much drug coverage they need, or opting for plans that have maximum annual or lifetime limits on drug coverage, or no longer providing coverage for retirees.[176] According to Ms. Anita Huberman, Chief Executive Officer, Surrey Board of Trade, “There are strains on all businesses. Costs are high and uncontrolled for those who do offer drug coverage. Costs are an impediment for some companies to offer any coverage.”[177]
E. Inappropriate PrescribingFinally, the Committee heard from witnesses that inappropriate prescribing practices are a critical issue facing the health care system. In her presentation, Dr. Anne Holbrook, Director, Division of Clinical Pharmacology & Toxicology, Professor, Department of Medicine, McMaster University, explained that high quality prescribing of prescription pharmaceuticals is difficult and complex with over 13,000 drugs on the Canadian market and an increasing number of seniors with multiple chronic diseases needing to take 10 or more medications simultaneously.[178] She explained that physicians lack the knowledge and skills necessary to deal with this increasingly complex situation, as only 9 to 50 hours of medical school is devoted to Clinical Pharmacology.[179] This void in training leaves physicians open to the influence of pharmaceutical companies, which may influence prescribing behaviour and promote the over prescribing of medications through face-to-face meetings with physicians, provision of samples and sponsoring educational and training events. As a result of inappropriate prescribing practices, the Committee heard that 300,000 Canadians suffer serious, disabling or fatal medication-related harm annually and 20% of Canadian medical malpractice cases relate to the prescribing of medications alone.[180]
While enhanced training and education is necessary to address this issue, the Committee heard from Drs. Martin and Holbrook that the creation of a national evidence-based drug formulary free of political and industry influence is critical. Dr. Cindy Forbes, then-President of the Canadian Medical Association, also explained that it is necessary for prescribers to have real-time information regarding what medications their patients are taking in order to prescribe drugs appropriately.[181] She explained that the federal government could support the development of e-prescribing through Canada Health Infoway Inc. The Canadian Pharmacists Association also explained that expanded pharmacy services such as medication reviews of prescriptions also play an important role in ensuring appropriate use and adherence to medications.[182] LESSONS LEARNED FROM OTHER JURISDICTIONSAs part of its study, the Committee also examined the implementation of national pharmacare programs in other jurisdictions to identify possible approaches that may be effective in a Canadian context. The Committee looked at pharmacare programs in Australia, New Zealand, Sweden and the Netherlands as they had been identified by witnesses as successful programs in terms of providing universal drug coverage and managing costs.[183] An overview of programs in these jurisdictions, as well as possible best practices that could be adopted in Canada is provided in the sections below. A. Australia’s Pharmaceutical Benefits SchemeAccording to Dr. David Henry, Professor, Dalla Lana School of Public Health, University of Toronto, Australia’s Pharmaceutical Benefits Scheme provides universal drug coverage to the entire population with affordable out-of-pocket expenditures.[184] Dr. Henry explained that Australia’s Pharmaceutical Benefits Scheme is codified in section 85 of the National Health Act. Under the scheme, the Therapeutic Goods Administration is responsible for assessing drugs for efficacy, quality and safety. The Pharmaceutical Benefits Advisory Committee then makes evidence-based recommendations to the federal minister of health about which drugs should be listed on the national evidenced-based drug formulary and provides advice on the price at which the drug represents value for money. The Pharmaceutical Benefits Pricing Authority negotiates drug prices based upon recommendations from the Pharmaceutical Benefits Advisory Committee. Once this negotiation is complete, the Minister adds the drug to the national formulary. A national prescribing service also provides education to family physicians regarding the use of medications. In evaluating Australia’s Pharmaceutical Benefits Scheme, Dr. Henry explained that the country is able to provide universal coverage at lower costs than Canada, spending $588 per capita compared to $771 in Canada in 2011, almost $200 less per person.[185] In addition to providing comprehensive coverage at lower costs, Dr. Henry explained that having a single system has permitted more efficient and economical bulk purchasing of expensive drugs for hepatitis C and has ensured that they are accessible to all sectors of the population, resulting in the disease’s complete eradication. According to Dr. Katherine Boothe at McMaster University, another benefit of the Australian model is that it integrates both formulary and drug pricing recommendations through the Pharmaceutical Benefits Advisory Committee.[186] However, Dr. Henry explained one drawback of the system is that the final drug listing decision remains up to the Minister of Health and is subject to cabinet approval, which means political and ideological considerations can play a role in drug listing decisions.[187] B. New Zealand’s Pharmaceutical Management AgencyPrescription drug coverage in New Zealand is provided through its national universal health care system, which is publicly funded through general taxation. The Pharmaceutical Management Agency (PHARMAC) is the government agency responsible for determining which drugs are covered on the Pharmaceutical Schedule, which includes both in- and out-of-hospital medications and medical devices.[188] Formulary listing decisions are based upon recommendations made by the Pharmacology and Therapeutics Advisory Committee, which undertakes a health technology assessment of prescription drugs. PHARMAC is also responsible for negotiating with manufacturers to establish prices and supply for medications on the Pharmaceutical Schedule based upon a fixed budget set by the Minister of Health.[189] Mr. Matthew Brougham explained to the Committee that New Zealand’s pharmacare program provides universal access with small co-payments ranging from NZ$0 to NZ$5 (C$4.44) per item per month.[190] He noted that though out-of-pocket payments do not exceed NZ$100 per year (C$88.80), they still can pose a barrier to individuals accessing pharmaceuticals. He further explained that the costs of the program have grown at a manageable rate of between 1% and 3% over the past 20 years. The drug formulary of the program covers over 2,000 drugs. He explained that it is difficult to compare New Zealand and Canada, but there are some lessons learned from New Zealand’s program that could be applicable to Canada. To manage costs, it is necessary to have a single drug purchasing organization with the power to negotiate a lower price. In addition, drug listing decisions need to be distanced from political decision-making to preserve negotiating power. Finally, the price for each drug needs to be set based upon the improvement to the quality of life it offers. In her appearance before the Committee, Ms. Heather Roy, Chair of the Board, Head Office, Medicines New Zealand, a pharmaceutical industry association, suggested that there are some challenges facing New Zealand’s system. In particular, she suggested that in her view PHARMAC is not transparent or timely in its decision-making in terms of drug listing decisions, which results in New Zealand lagging behind other countries in terms of access to medications.[191] However, Mr. Brougham explained that the drug listing decisions that took a greater amount of time were cases in which the price of the drug exceeded its benefits. It was therefore necessary for PHARMAC to negotiate a lower price with manufacturers, which takes a greater amount of time. Mr. Brougham further noted that “[t]hings that look like very good value go through very quickly. In my experience in the past, New Zealand was, in some instances, among the first countries to fund new technologies because it considered them to be very good value.”[192] Ms. Roy outlined other concerns her organization has with PHARMAC. She indicated that patients are forced to change medications once supply contracts negotiated by PHARMAC with drug manufacturers have ended or changed.[193] She therefore noted that it is important to ensure that the budget for a pharmacare program find an appropriate balance between choice and access and cost-savings. C. Sweden’s National Drug Benefits SchemeAccording to Ms. Sofia Wallström, Director General, Dental and Pharmaceutical Benefits Agency, Sweden provides universal drug coverage to its residents through the National Drug Benefits Scheme, which is financed through general taxation.[194] Levels of reimbursement are determined at the national level and are based upon the Act on Pharmaceutical Benefits, which outlines the three main principles of the program: the human value principle, the need and solidarity principle and the cost-effectiveness principle. Under the scheme, individuals cover the full costs of prescription drugs until they reach an annual threshold amount, after which they contribute co-payments until they reach a maximum annual out-of-pocket payment cap of SEK 2,200 (C$341) per year in 2017. The Dental and Pharmaceutical Benefits Agency is responsible for determining which drugs will be covered on the program’s drug formulary based upon an evaluation of their cost-effectiveness and their price is determined by their value to the health care system. In Sweden, regional or local governments referred to as county councils are responsible for setting and managing the local budget of the National Drug Benefits Scheme based upon financial agreements with the central government. Ms. Wallström explained to the Committee that Sweden is taking various approaches towards managing the rising costs of prescription pharmaceuticals.[195] First, generic substitution has been mandatory since 2002. In addition, pharmacies are required to dispense drugs with the lowest-price per unit. Further, patented medicines without a generic substitute face an automatic price cut of 7.5% after 15 years on the market. To address higher cost specialty drugs and biologics, county councils and drug manufacturers negotiate managed entry agreements, where reimbursement is dependent upon collection of data and demonstration of benefits. Finally, the Dental and Pharmaceutical Benefits Agency has also established a council composed of pharmaceutical companies and county councils to collaborate and have a national dialogue on determining pricing and reimbursement approaches for drugs for rare diseases. D. The NetherlandsMr. Aldo Golja, Senior Policy Advisor on Pricing and Reimbursement of Pharmaceuticals, Department of Pharmaceutical Affairs and Medical Technology, Dutch Ministry of Health, Welfare and Sports, explained that in the Netherlands, individuals are required by law to purchase private health insurance, but a minimum basket of services provided is established through legislation.[196] Therefore, all private insurances companies must provide the same broad health benefits package, which includes coverage for prescription pharmaceutical drugs. Out-of-pocket payments are capped annually at approximately C$574, including co-payments for pharmaceuticals. He explained that though maximum out-of-pocket payments in the Netherlands are relatively low, it is important to have mechanisms in place for individuals who cannot afford them to ensure that people take their medications.
The Dutch Health Institute is responsible for making drug formulary recommendations, based upon a cost-effectiveness evaluation of the drugs, or a health technology assessment. Prices are established through international reference prices, using prices in Belgium, the United Kingdom, Germany and France as comparators.[197] If there is more than one type of drug in a class offering comparable therapeutic effects, they use reference-based pricing, reimbursing the lowest priced drug in the class. The lowest priced drug in the class is reimbursed without a requirement for co-payment. Private insurance companies negotiate prices for generic drugs through tender contracts. They also negotiate contracts with health care providers, which include incentives for appropriate prescribing and use of generic drugs where ever possible. They also negotiate contracts with pharmacies for the delivery of their services. Managed entry agreements are negotiated for drugs for rare diseases. In these types of agreements, manufacturers agree to collect additional data regarding the drugs’ effectiveness and engage with providers to support appropriate use of the drugs in exchange for reimbursement of the drug. Coverage of biologics and biosimilars has been transferred to hospitals because they are able to negotiate lower prices for these drugs due to the volume of these drugs used in hospitals. Finally, Mr. Golja explained that international cooperation on price negotiation was an important component in addressing new high-priced specialty drugs, as they pose a challenge for all jurisdictions.[198] Throughout the Committee’s study, witnesses were very clear that Canada’s mix of public and private prescription drug coverage needs to be reformed in order to address gaps and variations in coverage, manage the rising costs of prescription pharmaceuticals, as well as improve overall health care. However, the question that now remains is how this reform can be achieved, given the complexity surrounding jurisdiction over pharmaceuticals and difficulties in managing rising costs. Possible policy options for expanding prescription drug coverage while managing prescription drug costs in Canada are outlined below, along with issues that need to be taken into account when moving forward in this area. A. Policy Options1. A Universal Public Prescription Drug Coverage ProgramIn their appearance before the Committee, Drs. Steven Morgan, Danielle Martin and Marc-André Gagnon, Associate Professor, School of Public Policy and Administration Carleton University, suggested that the establishment of a universal prescription drug coverage program is the best way to ensure that Canadians have equal access to prescription drugs, while ensuring that the drugs covered by the program offer value for money.[199] They outlined their recommendations, which are also presented in a report entitled Pharmacare 2020: The Future of Drug Coverage in Canada, to the Committee.[200] They recommended that the federal government, in collaboration with the provinces and territories, implement a public drug plan that provides universal coverage to all Canadians with little or no direct cost to patients. Dr. Martin explained that extremely low or zero co-payments are necessary because of the strong evidence that even very small co-payments can prevent individuals with relatively low incomes from filling their prescriptions.[201] To ensure that the pharmacare program is sustainable and offers value for money, it should only provide coverage for drugs listed on a single universal formulary, which would be developed based upon the best data and evidence regarding the risks and benefits of each drug.[202] The program would be managed by a publicly accountable management agency, which would be responsible for establishing the formulary and conducting joint price negotiations, combining the purchasing power of all provinces and territories and the federal government.[203] Dr. Marc-André Gagnon explained to the Committee that the new agency responsible for managing the pharmacare program could be modelled on Canadian Blood Services (CBS).[204] CBS is an independent agency funded and governed by provincial and territorial governments, which is responsible for bulk purchasing and managing a national formulary of plasma protein products developed in collaboration with the CADTH.[205] Dr. Gagnon explained to the Committee that a similar independent agency could be created to manage the pharmacare program by merging the CADTH and the pCPA.[206]Finally, Pharmacare 2020 proposes that the agency should have a predetermined annual budget that would be used to ensure that the money spent on pharmaceuticals maximizes health benefits.[207] Furthermore, this budget would be financed primarily by the provinces and territories, but the federal government would provide 25% of budget costs and operational expenses in line with its current level of contributions to the provinces and territories in support of the principles of the Canada Health Act.[208]
Dr. Steve Morgan told the Committee that the approach outlined in Pharmacare 2020 could result in savings of approximately $7 billion a year through reduced administrative costs, savings from joint price negotiations and drug purchasing, and reduced spending on drugs offering limited therapeutic benefit.[209] While a universal public drug coverage program would save money, it would also shift approximately $10 billion in costs from the private sector to the public sector.[210] According to the Pharmacare 2020 report, the federal government could raise funds for its portion of the agency’s budget through a variety of mechanisms including corporate taxes, income taxes, GST and/or premiums.[211] While the pharmacare program would supplant existing private drug coverage plans, private insurance companies could continue to play a role in administering claims for the program and/or providing additional coverage for drugs not listed on the formulary.[212] In his appearance before the Committee, Dr. Gregory Marchildon, Professor and Ontario Research Chair in Health Policy and System Design, Institute of Health Policy, Management and Evaluation, University of Toronto, further elaborated on the model presented in Pharmacare 2020, outlining two possible approaches regarding how a national universal public pharmacare program could be implemented in Canada.[213] For the first option, Dr. Marchildon explained that the national pharmacare program could follow the same approach as Medicare in Canada, which would involve expanding the Canada Health Act to include medically necessary prescription drugs dispensed outside of a hospital setting with federal financing provided through the Canada Health Transfer.[214] Provinces and territories would run their own single payer pharmacare plan and would remain legally responsible for their respective formularies. However, it would be possible for the federal, provincial and territorial governments to undertake negotiations to establish a single national drug formulary.[215] In addition, they could agree to the establishment of an intergovernmental agency, which would be responsible for managing the formulary and making formulary listing recommendations. However, the intergovernmental agency would remain voluntary policy body, rather than a law-making regulatory body and it would remain up to the provinces and territories to adopt its recommendations into law and/or regulations. Alternatively, to ensure compliance and national consistency, the federal government could instead make the national formulary a condition of eligibility for federal health transfers under the Canada Health Act.[216] Similarly, any provincial or territorial government refusing to adopt a recommendation of the intergovernmental agency could be subject to withdrawals of federal transfers.[217] In response to this proposal, Dr. Steve Morgan indicated that it was possible to move forward through the Canada Health Act, but it would require some changes.[218] Instead, he proposed creating an analogous Canada pharmacare act, which would “have the same purpose and maybe have more specific language about how this would be run, what kind of national agency would manage the formulary, how they would be subject to some budget constraint.”[219]
Finally, Dr. Marchildon proposed a second option in which the federal government would be entirely responsible for management and financing of the national universal pharmacare program.[220] Under the second option, prescription drug coverage would be provided to all Canadians by the federal government, replacing private and public coverage plans currently in place, with a single universal plan. Under this approach, there would be a federal formulary solely legislated and regulated by the federal government.[221] A federal agency would be responsible for determining the national formulary and undertake price negotiations with drug manufacturers. [222]According to Dr. Marchildon, this approach would offer the greatest potential to keep costs down, maintain clear lines of accountability, and eliminate individual and regional differences in coverage and access to prescription drugs.[223] However, he also noted that there are significant disadvantages in pursuing this approach, including “the lack of experience with such an approach, the fiscal risk that is assumed by the federal government alone, and the possibility that some provincial governments might reject the approach, despite the clear financial advantages of having this major cost pressure removed from their own budgetary responsibilities.”[224] 2. Reform the Current System of Private and Public Drug CoverageWhile many organizations[225] appearing before the Committee expressed support for the model proposed in Pharmacare 2020, others were concerned that a universal public drug insurance program would result in a significant shift in cost to governments and be mired in disputes between federal and provincial/territorial governments over funding.[226] Furthermore, the Committee heard that as many of the aspects related to the management of costs of such a program are under provincial jurisdiction, such as the prescribing practices of health care providers, a federal program may not make sense.[227] Consequently, these witnesses instead argued in favour of a more targeted approach towards addressing prescription drug coverage gaps, coupled with greater collaboration between the private and public sectors to control costs.[228] An overview of these proposals is provided below. According to Mr. Ake Blomqvist the federal government should focus on developing a strategy that takes action on aspects of pharmacare that it can undertake within its jurisdiction and supports reforms already underway at the provincial level.[229] To address gaps in prescription drug coverage, the federal government could ensure that every citizen has access to a default plan with an upper limit on the percentage of income that a family spends on drugs and offer partial financial support to provinces to meet that standard. Along these lines, the Canadian Medical Association proposed that as a possible first step towards universal coverage, the federal government could provide additional funding to the provinces and territories to allow them to deepen the coverage offered through their existing programs. The federal government could provide funding to the provinces and territories to enable them to establish an annual upper limit for out-of-pocket drug costs of $1,500 or 3% of income.[230] The Canadian Medical Association estimated that the cost to the federal government of covering the entire amount above the $1,500 or 3% threshold would be $1.6 billion in 2016.[231] To help manage costs and harmonize drug coverage across public and private plans, Mr. Blomqvist explained that the federal government could take the lead role in the pCPA and make arrangements to support the inclusion of private insurers in the pCPA.[232] In addition, patented drug prices could also be reduced substantially through regulatory changes to the PMPRB, as noted earlier in the report. Furthermore, public and private drug plans could collaborate to develop a common minimum national formulary for both public and private plans, as well as ensure consistent drug pricing, dispensing fees and additional mark-ups across drug plans.[233] Finally, public and private drug coverage plans could establish a common national approach towards the reimbursement of drugs for rare diseases.[234] B. Key Considerations in Moving ForwardIn examining the feasibility of these various policy proposals, witnesses highlighted key factors that should be taken into consideration when choosing an approach towards expanding prescription drug coverage in Canada, including potential costs and savings of a national pharmacare program; federal jurisdiction in relation to pharmaceuticals; financing arrangements; and impacts on the private sector; patients; federal client groups; and gender. 1. Potential Costs and Savings of a National Pharmacare ProgramOne of the main considerations raised by witnesses regarding the establishment of a national pharmacare program was its possible cost to federal, provincial and territorial governments.[235] While witnesses recognized the potential savings of such a program, as demonstrated by studies conducted by Drs. Steve Morgan, Marc-André Gagnon and others, they also identified the need for greater study of the costs and savings of such a program either by the federal government or the Parliamentary Budget Officer (PBO) to support its implementation.[236] Recognizing the importance of this issue, the Committee passed a motion on 29 September 2016 requesting that the PBO estimate the costs of creating and administering a single payer universal first-dollar prescription drug coverage program under the Canada Health Act.[237] This national pharmacare program would be financed through increased Canada Health Transfer payments to provinces and territories to cover the costs of expanding their existing programs. In developing its terms of reference for the study, the Committee agreed that the proposed national pharmacare program would provide coverage for the drugs currently listed on the Province of Quebec’s Public Prescription Drug Insurance Plan (RAMQ) List of Medications to allow for the PBO to estimate the potential costs of the program. This formulary was chosen because it is broad, covering 8,000 out of the 13,000 available drugs in Canada, including exceptional medications such as cancer drugs dispensed outside hospitals. The formulary also serves as the basis of coverage for both public and private plans in Quebec.[238] In line with witness testimony regarding potential challenges posed by co-payments, the Committee agreed that co-payments would be limited to $5 per brand-name drug listed on the formulary and there would be no co-payments for generic drugs. However, some populations would be exempt from co-payments altogether, including individuals 15 and under, seniors, pregnant women, individuals with low incomes and disabilities, and students between 16 and 18 years of age. On 17 October 2017, the PBO presented the findings of its report entitled Federal Cost of a National Pharmacare Program to the Committee.[239] In its costing analysis, the PBO found that under a national pharmacare program, total prescription drug spending per 2015-2016 figures based upon the Quebec formulary would amount to $20.4 billion (see figure 11).[240] This takes into account that the PBO estimates that the creation of a national pharmacare program would increase drug consumption by 12.5%, resulting in an increase of total drug expenditure by $1.7 billion, which would be more than offset by a $5.9 billion decrease, resulting from:
Finally, the PBO estimated that the costs of the program would have a compound growth rate of 3.1% per year, resulting in gross costs of the program reaching $23.7 billion in 2020-2021.[242] Figure 11. Costs and Savings of a National Pharmacare Program
Under the PBO considered scenario, public insurance plans would absorb out-of-pocket costs from individuals ($ 4.7 billion) except for revenue from co-payments ($397 million), resulting in a net savings to individuals of $4.3 billion.[243] As a result, individual out-of-pocket expenditures for prescription drugs would be reduced by 90% on average. Individuals of working age would obtain 69% savings, while individuals who are exempt from co-payments, such as seniors, children, students and individuals with disabilities and those receiving income assistance, would obtain 100% savings in out-of-pocket expenses. Similarly, current private insurance plan spending would be reduced by $10.7 billion, which would also be absorbed by public insurance plans. Accounting for the noted co-payments and the absorption of current private sector costs, the total cost to the public system would then be $20.0 billion in 2015-2016, marking a net incremental increase in spending by public insurance plans of $6.9 billion (see Table 2 for further details). Table 2. Costs of a National Pharmacare Program by Primary Payer, 2015-2016
Source: Adapted from Table 3-6 in Office of the Parliamentary Budget Officer, Federal Cost of a National Pharmacare Program, 28 September 2017, p. 42. In his appearance before the Committee, the Parliamentary Budget Officer explained that the PBO’s analysis represented a prudent and balanced estimate of costs.[244] The report noted that implementation of a national pharmacare program may result in additional savings from reductions in administrative costs from the elimination of private plans. These costs were estimated to be 5.7% or $3.6 billion of total private sector drug costs in 2014.[245] He also noted that joint price negotiations may result in additional savings of up to 30%.[246] Ms. Carleigh Malanik, Financial Analyst, Office of the Parliamentary Budget Officer, also indicated that the costing did not make any assumptions about whether there would be changes to policies related to mark-ups and pharmacists’ fees, which were kept at 2017 rates and amounted to $7.4 billion of the total program cost.[247]
Though difficult to estimate exact amounts, the Canadian Centre for Policy Alternatives noted, in a brief submitted to the Committee, that there may also be additional indirect savings from a reduction of health care expenditures arising from individuals who experience health complications from not taking their medications due to cost.[248] It also pointed out that a significant proportion of private sector costs are already indirectly assumed by different levels of governments through their provision of drug coverage to public sector employees. They estimated this amount to be $3.34 billion in 2016, funding which could be redirected towards a new pharmacare program.[249] Finally, Dr. Marc-André Gagnon noted that the PBO’s report did not examine the possible reduction of public spending from the removal of federal tax subsidies for private insurance plans or possible impacts of the program on federal medical tax credits.[250] 2. Jurisdictional IssuesThe Committee heard from witnesses that jurisdictional issues are another key consideration in identifying the best approach towards developing a national pharmacare program in Canada. The Committee heard from legal experts that the use of the federal government’s spending power is the most feasible approach for expanding prescription drug coverage to all Canadians.[251] This approach could involve expanding the Canada Health Act to include prescription drugs dispensed outside of hospitals as an insured service under the Act. However, Professor Bruce Ryder explained that there are limits to the federal government’s expenditure power in terms of the conditions it can place on providing funding to the provinces.[252] If the federal government were to require the provinces and territories to provide coverage for a defined list of medications, or a national formulary under the Canada Health Act, the specificity of this particular requirement might be considered as going beyond federal jurisdiction, as it could be considered the regulation of the delivery of health services which is a provincial matter. [253] Currently, the Canada Health Act does not define which specific physician or hospital services are considered to be medically necessary in order for them to be covered by a provincial health insurance plan under the Act.[254] Rather, the provinces and territories in consultation with medical bodies remain responsible for determining which specific physician and hospital services are considered medically necessary and will be covered under their respective provincial and territorial health insurance plans.[255] Professor Colleen Flood, Director of the Centre for Health Law, Policy and Ethics, University of Ottawa explained that the federal government could instead include a provision in the Act that requires the provinces and territories to have a fair and transparent process for deciding which drugs would be covered, but provinces and territories would remain responsible for managing the formulary.[256] A national drug formulary could be developed collaboratively by the federal, provincial, and territorial governments but its adoption should remain voluntary, rather than a specific requirement under the Canada Health Act.[257]
The Committee heard that the imposition of a single drug pricing and purchasing organization by the federal government could extend beyond Parliament’s jurisdiction over patents and may be subject to legal challenge. Witnesses explained that a similar approach was taken to establish a national securities regulator under the Securities Act.[258] Despite having opt-out provisions for the provinces, the Supreme Court of Canada found that the federal government’s establishment of a national securities regulator could not be supported under its power over trade and commerce under section 91(2) of the Constitution Act, 1867.[259] However, Professor Amir Attaran explained the court’s decision would allow for the provinces and territories to cooperate through private contracts to jointly purchase medications, possibly designating a common buying agent.[260] Alternatively, as explained by Dr. Marchildon, earlier in this report, federal, provincial and territorial governments could negotiate the establishment of an intergovernmental agency that would be responsible for making policy recommendations regarding drug formulary listing decisions.[261] However, under this approach, the decision by provinces and territories to adopt its recommendations into law and/or regulation would remain voluntary. While moving forward towards a national pharmacare program could pose jurisdictional challenges, the Committee heard that it is necessary for the federal government to play a leadership role in overcoming these challenges and promoting collaboration among the provinces and territories to address the health needs of all Canadians, as it did with the establishment of Medicare in the 1960s.[262] Dr. Katherine Boothe emphasized that it is not necessary for the federal government to have sole responsibility for the national formulary or price negotiation process, as provinces and territories collaborate well through the cPCA and the CADTH. Rather, it is necessary for the federal government to act as a crucial partner by providing financial incentives for the provinces and territories to commit to common standards related to the provision of prescription drug coverage in Canada, an approach that is consistent with the provision of other medically necessary services under the Canada Health Act.[263]
3. Financing ArrangementsAs outlined in the PBO’s report, the establishment of a universal single payer prescription drug coverage program would require public prescription drug plans to assume an additional $7.2 billion in spending from private health insurance providers.[264] Witnesses appearing before the Committee identified the need to examine possible ways federal, provincial and territorial governments could raise funds to finance the expansion of public prescription drug coverage to all Canadians. In his appearance before the Committee, Professor Steven Morgan explained that a new revenue tool would have to be created to raise funds to finance the program.[265] The purpose of this revenue tool would be to redirect money currently spent by the private sector on prescription drug coverage towards the public program: What we need is to find a revenue tool to move some of the money that is in the private sector—some of the money for publicly financed private drug benefits for people like me, a public employee—into the system. We don't need new money in Canada to run a pharmacare system; we just need a new tool to move the money we're already spending into the system so that it functions more equitably and efficiently.[266] Witnesses outlined some possible revenue tools that could be used to redirect spending from the private sector to the public program. Dr. Marc-André Gagnon explained that revenue could be raised through an increase in corporate taxes, which is a tax on the profit income and capital of corporation. [267] This increase in corporate taxes would be offset by employers having lower labour costs as a result of the creation of the new public program.[268] Alternatively, he indicated that funds could be raised through a payroll tax, which is a common approach for funding social security programs.[269] A payroll tax is a tax levied on an employee’s wage or salary and is collected by the employer through payroll deductions. In the context of the funding of social insurance programs, a payroll tax is usually paid for entirely or in part by the employer based upon a percentage of their total salaries or wages paid.[270] If the payroll tax is not entirely paid for by the employer, the employee also faces a deduction to their wage or salary. Dr. Gagnon indicated that raising funds through general taxation is also a possible approach.[271] Dr. Colleen Flood explained that premiums are also a means of financing the program and are permissible under the Canada Health Act, as some provinces currently charge premiums under the Act for health services. However, these are generally paid for indirectly through income taxes and/or payroll taxes to avoid financial barriers to accessing care.[272] Finally, witnesses were in agreement that out-of-pocket payments for prescription pharmaceuticals in the form of co-payments, premiums or deductibles should not be used as a primary means of financing the program. As Dr. Monika Dutt pointed out, even the smallest co-payments amounting to $10 could result in individuals not taking their medications.[273] In examining support among Canadians regarding different approaches towards financing a pharmacare program, Ms. Shachi Kurl indicated that the majority of respondents to their 2015 survey were in favour of raising corporate federal tax levels.[274] However, there was limited support for increases in the GST, basic income taxes or charging of an annual premium of $180.[275] In terms of cost-sharing arrangements between different levels of government, the Committee heard that costs for expanding prescription drug coverage should be shared between the federal, provincial and territorial governments, given that costs for health care delivery are also currently shared.[276] 4. Impacts on the Private SectorAccording to Dr. Marc-André Gagnon, a survey by Benefits Canada indicated that 53% of employers would be in favour of a public drug coverage plan.[277] Similarly, the 2015 Angus Reid Survey found that an overwhelming majority of Canadians (91% of respondents) were also in favour of a national pharmacare program.[278] Ms. Anita Huberman, Chief Executive Officer, Surrey Board of Trade, said that businesses would support a national public pharmacare program, but must be at the table in negotiating funding mechanisms.[279] Ms. Dianne Balon, Vice-President, Government, Alberta Blue Cross, indicated that any changes to private drug coverage should consider implications for the coverage level of other supplementary health benefits offered by employers, such as dental and vision benefits and mental health.[280] Ms. Huberman further explained that employers could use the savings that they obtain from the creation of a national pharmacare program to enhance their coverage of other services, such as mental health, vision care, dental care, hearing care, and physiotherapy.[281] However, she noted that the coverage offered by the public program would have to be comprehensive, as limited prescription drug coverage programs such as British Columbia’s Catastrophic Drug Coverage program, continue to pose burdens on employers by not meeting employee health needs.[282] According to CUPE, drug benefits are a contentious and difficult issue for unions to negotiate. Labour unions are therefore supportive of no longer being responsible for determining the level and type of drug coverage that their members should have.[283] The Canadian Life and Health Insurance Association pointed out that there would be transactional costs and challenges associated with shifting patients from their current medications to those covered by a public plan formulary.[284]
5. Patient PerspectivesThe Committee heard that patient groups[285] are supportive of expanding prescription drug coverage to all Canadians, as well as establishing national standards for coverage across the country. They did not however advocate for a particular model of pharmacare. They expressed concern that a national formulary could potentially limit patient choice and stressed the need for patients to be at the table in developing new approaches to providing prescription drug coverage. The Auditor General of Canada further pointed out that it is important for public programs to be designed around the needs of citizens so they can easily access and navigate them.[286] 6. Needs of Federal Client GroupsThe Committee heard from federal officials responsible for administering drug coverage programs for First Nations and Inuit, as well as veterans that if a national pharmacare program were to be created there would be a need to ensure that it meets the needs of different population groups. For example, First Nations and Inuit would need to have access to forms of therapies that can be delivered in rural and remote areas.[287] Current formulary listing recommendations made by the CADTH reflect the needs of populations living in urban or suburban areas. Further, Mr. Sony Perron explained that it would be important to take into consideration governance arrangements with First Nations and Inuit peoples, as the Department is currently working with the AFN to support First Nations and Inuit having greater control over their own programs as part of a joint review of the NIHB program.[288] 7. Gender ImpactsIn a written submission to the Committee, Dr. Cara Tannenbaum, Scientific Director, Institute of Gender and Health, Canadian Institutes of Health Research explained that it is necessary to apply sex and gender-based analysis to the development and evaluation of decisions related to drug pricing, drug access, and the development of a national drug formulary.[289] According to the submission, drug pricing and coverage decisions have different impacts on women and men, as per capita spending on drugs differs by gender. In addition, women often need lower doses of drugs, which could have implications for drug pricing. In addition, women also experience higher rates of adverse drug events, which could have implications for drug approvals. Dr. Tannenbaum’s written submission also highlighted the need to ensure that drug formulary listing decisions support access to a range of hormone-related contraceptive options to promote gender equity.[290] Finally, expansion of public drug coverage to all Canadians is likely to improve women’s access to prescription pharmaceuticals, as they are less likely than men to have access to employer-based coverage because they are more likely to occupy part-time positions where drug benefits are not offered by employers.[291] Furthermore, income gaps between men and women mean that out-of-pocket payments for prescription drugs pose a greater burden on women, particularly female seniors with low incomes.[292] The Committee recognizes the critical importance that prescription drugs play in improving health outcomes for Canadians. However, the Committee’s study has demonstrated that Canada does a poor job of making sure that prescription drugs are accessible to all Canadians. Over the years, there have been incremental improvements to Canada’s mix of public and private drug coverage plans through the offering of catastrophic drug plans or more general public plans. Federal, provincial and territorial governments are collaborating more closely than ever to negotiate joint prices for prescription pharmaceuticals. However, these incremental reforms have not benefited all Canadians. Current public drug plans still leave many Canadians uninsured, while savings from joint price negotiations do no benefit individuals who lack insurance, or the 70% of Canadians who obtain coverage through private plans. What are the results of these incremental reforms? Too many Canadians must still choose between the necessities of life and filling their prescriptions. Too many employers must now choose between laying off their employees and limiting the drug benefits they provide. Incremental improvement is no longer enough. The majority of the Committee believes that it is time to move forward and create a universal single public payer prescription drug coverage program for all Canadians. Given our federated state, the Committee believes that this program should be delivered collaboratively by federal, provincial and territorial governments. The Committee believes that the best approach for the creation of such a program is through the expansion of the Canada Health Act to include prescription drugs dispensed outside of hospitals as an insured service. The Committee also believes that the program should be cost-shared between federal, provincial and territorial governments. The program would also include the development of a national voluntary prescription drug formulary through collaboration between federal, provincial and territorial governments, health care providers, patients and Indigenous communities that would help guide reimbursement decisions and promote consistency in drug coverage listing decisions across the country. As the main purpose of the creation of a universal single public payer prescription drug coverage program is to ensure that no Canadian faces financial barriers in accessing medically necessary prescription drugs, the Committee strongly discourages the use of co-payments as means of financing the program. However, the Committee believes that if co-payments are used, they should be structured to promote the appropriate and cost-effective use of medications, such as promoting generic substitution for brand name drugs, where possible. In no case should they be used as a barrier to care. The Committee recognizes that in moving towards universal publicly funded prescription drug coverage, governments will be assuming significant costs that are currently borne by the private sector in the order of $10.7 billion before potential savings are applied. To realize the $4.2 billion in savings that universal drug coverage will provide, it is necessary to expand and build capacity within the CADTH and the pCPA to support the development of a pan-Canadian formulary and more robust price negotiations. While it may be necessary from a constitutional perspective for these approaches to remain voluntary, the Committee hopes that significant investments by the federal government will enable provinces and territories to commit to common standards for prescription drug coverage. Though voluntary, a defined national drug formulary will also empower patients to hold their governments to account in ensuring that they have access to those drugs. Finally, the Committee recognizes that its study only grazed the surface in terms of identifying possible approaches towards raising federal funds to support the expansion of the Canada Health Act. The Committee heard that it would be necessary for the federal government to undertake consultations with employers, unions, private plans and Canadians more broadly to identify possible approaches towards financing a national pharmacare program, as well as re-evaluate its current tax measures that support the provision of private health insurance. The Committee notes that since the completion of its study, the PMPRB has released new regulations on 2 December 2017 that will strengthen its ability to regulate the prices of patented medications through the adoption of value-based pricing; changes to the countries that it uses for price comparisons; and the introduction of new reporting requirements on price rebates offered by manufacturers to third parties.[293] The organization estimates that these regulatory changes will result in $12.6 billion in savings over 10 years.[294] The Committee welcomes these developments and believes that these savings create additional fiscal room that can be used by governments to expand prescription drug coverage to all Canadians through a national pharmacare program under the Canada Health Act. A national pharmacare program would also ensure that all Canadians benefit from these identified savings. While leadership on the part of governments is necessary to ensure Canadians have access to prescription medications, the Committee’s study also highlighted that health care providers have a critical role to play in ensuring that the medications they prescribe are appropriate and do not result in adverse health outcomes. From the raging opioid crisis to the slow moving tsunami of antimicrobial resistance, the Committee has heard that health care providers must improve their prescribing practices. As self-regulating professions, governments and Canadians have entrusted health care providers to act as stewards of the health care system. However, it is also necessary for governments to provide them with the tools and supports to do their job effectively, including such as through the proposed national formulary. In recognition of the federal minister of health’s mandate to “improve access to necessary prescription medications,”[295] the Committee makes the following recommendations: RECOMMENDATIONSA. Expanding the Canada Health Act to include Prescription Drugs Dispensed Outside Hospitals In order to implement a single payer universal prescription drug coverage program for all Canadians, the Committee recommends: Recommendation 1 That the Government of Canada work in collaboration with provinces and territories, health care providers, patients and Indigenous communities to develop a common voluntary national prescription drug formulary. Recommendation 2 That the Government of Canada amend the Canada Health Act to include drugs prescribed by a licensed health care practitioner and dispensed outside of hospitals in accordance with a common voluntary national formulary, as part of the definition of an “insured health service” under the Act. Recommendation 3 That the Government of Canada provide additional funding to provinces and territories through the Canada Health Transfer to support the inclusion of prescription drugs dispensed outside of hospitals as an insured service under provincial and territorial public health insurance programs under the Canada Health Act. Recommendation 4 That the Government of Canada undertake consultations with employers, unions, private plans and Canadians at large to identify possible approaches towards financing the expansion of the Canada Health Act to include prescription drugs dispensed outside of hospitals as an insured service. Recommendation 5 That the Government of Canada undertake consultations with First Nations and Inuit communities to determine whether it is their preference to obtain prescription drug coverage under the Canada Health Act or through the Non-Insured Health Benefits Program, with the ultimate goal of recognizing the authority of First Nations and Inuit peoples in providing health services to their communities. B. Development of a Common Voluntary National Prescription Drug Formulary The Committee recommends: Recommendation 6 That the Government of Canada apply gender-based plus analysis in the development of the common voluntary national prescription drug formulary. Recommendation 7 That the Government of Canada share the costs of the prescription drugs listed on the common voluntary national formulary and associated professional fees with the provinces and territories through the Canada Health Transfer. C. Improving Drug Pricing and Reimbursement Processes The Committee recommends: Recommendation 8 That the Government of Canada, in collaboration with the provinces and territories, expand the mandate of the Canadian Agency for Drugs and Technologies in Health to require it to maintain the common national voluntary prescription drug formulary and provide guidance to health care providers to support its use. Recommendation 9 That the Government of Canada provide the Canadian Agency for Drugs and Technologies in Health with additional funding to expand its capacity to undertake therapeutic reviews of high cost specialty drugs, oncology drugs and drugs for rare diseases, as well as develop expertise to support the negotiation of managed entry agreements for these drugs. Recommendation 10 That the Government of Canada, in collaboration with the provinces and territories, develop a transparent decision-making framework for price negotiations for pharmaceutical drugs undertaken by the pan-Canadian Pharmaceutical Alliance based upon best practices. Recommendation 11 That the Government of Canada, in collaboration with the provinces and territories, designate the pan-Canadian Pharmaceutical Alliance as the common agent for the bulk buying of prescription drug pharmaceuticals. Recommendation 12 That the Government of Canada align the mandate of the Patented Medicine Prices Review Board with the policies and priorities of the Canada Agency for Drugs and Technologies in Health. Recommendation 13 That the Government of Canada amend the Patent Act and/or establish regulations requiring that patented drug manufacturers reduce their prices after 15 years, if no generic substitute for a patented prescription drug is available, in line with practices in other jurisdictions. Recommendation 14 That the Government of Canada investigate the market practices of the pharmaceutical sector, including those of patented and generic drug manufacturers, wholesalers and retail pharmacies to identify opportunities to promote price reductions of prescription drugs through greater competition. Recommendation 15 That the Minister of Health enter into discussions with provincial and territorial counterparts with the aim of reducing the delays in access to new non-prescription medicines by integrating the drug scheduling process into the federal non-prescription drug approval process. D. Improved Data and Information Systems The Committee recommends: Recommendation 16 That the Government of Canada collaborate more closely with the provinces, territories and the private sector to accelerate the development of a complete national data system on the utilization of prescription pharmaceuticals in Canada to support the management of prescription drug coverage programs in Canada. Recommendation 17 That Health Canada, Canadian Institute for Health Information and Canada Health Infoway Inc. collaborate to develop a national real-time electronic adverse drug reaction reporting system. Recommendation 18 That the Government of Canada request that the Office of the Parliamentary Budget Officer make the data commissioned for their study of the federal cost of a new national pharmacare program available to the public and other government agencies. [1] House of Commons Standing Committee on Health (HESA), Evidence, 1st Session, 42nd Parliament, 20 April 2016, 1640 (Dr. David Henry, Professor, Dalla Lana School of Public Health, University of Toronto, As an Individual). [2] Ibid. [3] Angus Reid Institute, “Canadian Public Opinion Regarding a National Pharmacare Program,” written submission to HESA, 1st Session, 42nd Parliament, 6 June 2016. [4] CIHI data tables: How Canada compares: results from the Commonwealth Fund’s 2016 International Health Policy Survey of Adults in 11 countries, submitted by Dr. Marc-André Gagnon, Associate Professor, School of Public Policy and Administration, Carleton University. [5] Office of the Parliamentary Budget Officer (PBO), Federal Cost of a National Pharmacare Program, 28 September 2017. [6] Express Scripts Canada, “Drug Trend Report, 2016,” p. 24. [7] HESA, Minutes of Proceedings, 1st Session, 42nd Parliament, 7 March 2016. [8] HESA, Minutes of Proceedings, 1st Session, 42nd Parliament, 29 September 2016. [9] PBO, Federal Cost of a National Pharmacare Program, 28 September 2017. [10] Ms. Karin Phillips, Catastrophic Drug Coverage in Canada, Parliamentary Information and Research Service, Library of Parliament Publication, Background Paper No. 2016-10-E, 4 February 2016. [11] For a definition of patent, please see: Government of Canada, What is a Patent?. [12] PBO, Federal Cost of a National Pharmacare Program, 28 September 2017, p. 10. [13] Ibid. [14] Ms. Virginie Demers et al., “Comparison of provincial prescription drug plans and the impact on patients’ annual drug expenditures,” Canadian Medical Association Journal, Vol. 178, No. 4, 12 February 2008. [15] PBO, Federal Cost of a National Pharmacare Program, 28 September 2017, p. 18. [16] Constitution Act, 1867, 30 & 31 Victoria, c. 3 (U.K.). [17] HESA, Evidence, 1st Session, 42nd Parliament, 23 February 2017, 1105 (Professor Bruce Ryder, Associate Professor, Osgoode Hall Law School, York University, As an Individual). [18] HESA, Evidence, 1st Session, 42nd Parliament, 13 April 2016, 1545 (Ms. Abby Hoffman, Assistant Deputy Minister, Strategic Policy Branch, Department of Health). [19] Patented Medicine Prices Review Board (PMPRB), Annual Report, 2016. [20] Ibid. [22] Canada Health Act, R.S.C., 1985, c. C-6, s. 7. [23] Ibid., s. 2 [24] PBO, Federal Cost of a National Pharmacare Program, 28 September 2017, p. 17. [25] Ibid. [26] Ibid. [28] Ibid. [29] Ibid., 1130. [30] HESA, Evidence, 1st Session, 42nd Parliament, 13 April 2016, 1610 (Dr. Brian O’Rourke, President and Chief Executive Officer, Canadian Agency for Drugs and Technologies in Health (CADTH)). [31] Ibid. [32] Ibid. [33] Ibid. [34] HESA, Evidence, 1st Session, 42nd Parliament, 2 May 2016, 1550 (Mr. William Dempster, Chief Executive Officer, 3Sixty Public Affairs). [37] Ibid. [38] Canada’s Premiers, pan-Canadian Pharmaceutical Alliance. [39] Canadian Institute for Health Information (CIHI), About CIHI. [40] HESA, Evidence, 1st Session, 42nd Parliament, 13 April 2016, 1555 (Mr. Brent Diverty, Vice President of Programs, CIHI). [41] Ibid. [43] PBO, Federal Cost of a National Pharmacare Program, 28 September 2017, p. 17. [44] HESA, Evidence, 1st Session, 42nd Parliament, 13 April 2016, 1545 (Hoffman) and PBO, Federal Cost of a National Pharmacare Program, 28 September 2017, pp. 1, 27. [45] PBO, Federal Cost of a National Pharmacare Program, 28 September 2017, p. 1. [46] Ibid., p. 27. [47] Ms. Karin Phillips, Catastrophic Drug Coverage in Canada, Background paper no. 2016-10-E, Parliamentary Information and Research Service, Library of Parliament, Ottawa, 4 February 2016. [48] Ibid. [49] Régie de l’assurance maladie du Québec (RAMQ), Prescription drug insurance. [50] RAMQ, Prescription drugs covered. [51] HESA, Evidence, 1st Session, 42nd Parliament, 2 February 2017, 1120 (Ms. Dianne Balon, Vice-President, Government of Alberta Blue Cross). [52] The New Brunswick Drug Plan, Information Sheet. [53] Government of Prince Edward Island, Apply for the Generic Drug Program. [54] Unless otherwise noted, this section is based upon the following document: Ms. Karin Phillips, Catastrophic Drug Coverage in Canada, Background paper no. 2016-10-E, Parliamentary Information and Research Service, Library of Parliament, Ottawa, 4 February 2016. [55] HESA, Evidence, 1st Session, 42nd Parliament, 1 December 2016, 0845 (Mr. Sony Perron, Senior Assistant Deputy Minister, First Nations and Inuit Health Branch, Department of Health). [56] Since the completion of the Committee’s study, the First Nations and Inuit Health Branch and the administration of the NIHB program has been transferred to the Department of Indigenous Services by Order in Council 2017-1465 on 29 November 2017. [57] Ibid. [58] Ibid. [59] Ibid. [60] HESA, Evidence, 1st Session, 42nd Parliament, 1 December 2016, 0900 (Mr. Michel Doiron, Assistant Deputy Minister, Service Delivery Branch, Department of Veterans Affairs). [61] Ibid. [62] HESA, Evidence, 1st Session, 42nd Parliament, 2 February 2017, 1105 (Commander Sylvain Grenier, Senior Staff Officer, Pharmacy Services, Department of National Defence). [63] Ibid. [64] Government of Canada, Interim Federal Health Program: Summary of Coverage. [65] Ibid. [66] Government of Canada, Public Service Health Care Plan at a glance. [67] Correctional Service Canada, 2017–18 Departmental Plan. [68] Canadian Union of Public Employees (CUPE), “Development of a National Pharmacare Program,” written submission to HESA, 1st Session, 42nd Parliament, September 2016. [70] Canadian Life and Health Insurance Association, “Canadian Life and Health Insurance Industry Facts, 2017 eds.,” 2017. [71] Ibid. [72] PBO, Federal Cost of a National Pharmacare Program, 28 September 2017, p. 18. [73] Ibid. [74] Ibid., p. 17. [75] CIHI, “National Health Expenditure Trends, 1975-2017,” p. 15. [76] Ibid., p. 18 and CIHI, “Prescribed Drug Spending in Canada, 2017: A Focus on Public Drug Programs,” 2017, p. 7. [77] CIHI, “Prescribed Drug Spending in Canada, 2017: A Focus on Public Drug Programs,” 2017, p. 7. [78] Ibid., p. 8. (Non-drug medical products include items related to the delivery of prescription drugs such as diabetic supplies). [80] PMPRB, “Annual Report, 2016,” 2017, p. 49. [81] CIHI, “Prescribed Drug Spending in Canada, 2017: A Focus on Public Drug Programs,” 2017, p. 7. [82] PBO, Federal Cost of a National Pharmacare Program, 28 September 2017, p. 42. [83] CIHI, “Prescribed Drug Spending in Canada, 2017: A Focus on Public Drug Programs,” 2017, p. 27. [84] PBO, Federal Cost of a National Pharmacare Program, p. 7. [85] Ibid., p. 15. [86] Ibid., p. 6. [87] Federal Cost of a National Pharmacare Program, p. 20. The PBO noted in its report that there are significant variations in how both CIHI and QuintilesIMS determine out-of-pocket costs for individuals, resulting in a variation of $2.4 billion in the amount of out-of-pocket costs between the different data sets. CIHI reported $7.1 billion in out-of-pocket expenses in 2015-2016, whereas QuintilesIMS reported $4.7 billion. The PBO’s report uses QuintilesIMS data for out-of-pocket expenditures, where primary payer refers to the payer, public insurance, private insurance or individual out-of-pocket that paid for the largest portion of the prescription. The entire value of the drug is attributed to the primary payer, even though this out-of-pocket amount may be reimbursed by an insurer as through a co-ordination for benefits. For further explanation regarding data limitations in relation to out-of-pocket costs, please see, PBO, p. 20. [88] HESA, Evidence, 1st Session, 42nd Parliament, 13 April 2016, 1555 (Diverty and Ms. Tanya Potashnik, Director, Policy and Economic Analysis Branch, PMPRB). [89] Ibid., 1605 (Potashnik). [90] Health Canada’s draft definition of a rare disease is one that affects fewer than one in 2,000 persons in Canada. HESA, Evidence, 1st Session, 42nd Parliament, 30 May 2016, 1535 (Dr. Durhane Wong-Rieger, President and Chief Executive Officer, Canadian Organization for Rare Disorders). [91] Ibid., 1555 (Dr. Christopher McCabe, Capital Health Research Chair, Faculty of Medicine and Dentistry, University of Alberta). [92] CIHI, Prescribed Drug Spending in Canada, 2017: A Focus on Public Drug Programs, 2017, p. 6. [93] Ibid., p. 26. [94] PBO, Federal Cost of a National Pharmacare Program, p. 18. [95] Express Scripts Canada, “Drug Trend Report, 2016,” p. 9. [97] Ibid. (Potashnik). [98] PMPRB, Generic Drugs Canada, 2015. [99] PMPRB, “Annual Report, 2016,” 2017, p. 32. The foreign-to-Canadian price ratio is a comparison of the national average price of a drug in Canada, based on all manufacturer ex-factory sales in the retail sector, compared to the sales-weighted national average in another country. For the purposes of analysis, the Canadian patented drug price is set to a value of one and the foreign price is expressed as a proportion of the Canadian price. An average foreign-to-Canadian price ratio below one indicates a lower average foreign price, while a ratio above indicates a higher price. [100] HESA, Evidence, 1st Session, 42nd Parliament, 13 April 2016, 1555 (Diverty) and CIHI, “Prescribed Drug Spending in Canada, 2017: A Focus on Public Drug Programs — International Comparisons, Data Tables,” 2017. [101] HESA, Evidence, 1st Session, 42nd Parliament, 6 June 2016, 1555 (Mr. Glenn Monteith, Vice-President, Innovation and Health Sustainability, Innovative Medicines Canada). [102] Ibid. [104] Ibid. [105] HESA, Evidence, 1st Session, 42nd Parliament, 27 September 2016, 0905 (Mr. Victor Elkins, Regional Vice-President for British Columbia, Canadian Union of Public Employees (CUPE)). [106] CUPE, “Development of a National Pharmacare Program,” written submission to HESA, 1st Session, 42nd Parliament, September 2016. [107] HESA, Evidence, 1st Session, 42nd Parliament, 18 April 2016, 1540 (Dr. Danielle Martin, Vice-President, Medical Affairs & Health System Solutions, Women’s College Hospital). [108] CUPE, “Development of a National Pharmacare Program,” written submission to HESA, 1st Session, 42nd Parliament, September 2016. Wellesley Institute, “Low Earnings, Unfilled Prescriptions: Employer-Provided Health Benefit Coverage in Canada, July 2015,” p. 8 reference document cited by CUPE . [109] Ibid. [111] HESA, Evidence, 1st Session, 42nd Parliament, 1 June, 2016, 1535 (Ms. Natasha Mistry, Director, Stakeholder Relations and Community Development, Canadian Association of Retired Persons). [112] PBO, Federal Cost of a National Pharmacare Program, p. 21. [113] Ibid. [114] HESA, Evidence, 1st Session, 42nd Parliament, 6 June 2016, 1530 (Ms. Shachi Kurl, Executive Director, Angus Reid Institute). [115] HESA, Evidence, 1st Session, 42nd Parliament, 22 September 2016, 0935 (Dr. Jan Hux, Chief Science Officer, Canadian Diabetes Association). [116] HESA, Evidence, 1st Session, 42nd Parliament, 16 May 2016, 1600 (Ms. Connie Côté, Executive Director, Health Charities Coalition of Canada). [117] HESA, Evidence, 1st Session, 42nd Parliament, 6 June 2016, 1630 (Dr. Monika Dutt, Chair, Canadian Doctors for Medicare). [118] Ibid., 1600. [119] HESA, Evidence, 1st Session, 42nd Parliament, 18 April 2016, 1530 (Dr. Steven Morgan, Professor, School of Population and Public Health, University of British Columbia, As an Individual). [121] HESA, Evidence, 1st Session, 42nd Parliament, 1 June 2016, 1545 (Dr. Cindy Forbes, President, Canadian Medical Association); and HESA, Evidence, 1st Session, 42nd Parliament, 30 May 2016, 1535 (Wong-Rieger). [124] Assembly of First Nations (AFN), The First Nations Health Transformation Agenda, February 2017. [125] Ibid., p. 97. [129] HESA, Evidence, 1st Session, 42nd Parliament, 9 May 2016, 1600 (Mr. Frank Swedlove, President and Chief Executive Officer, Canadian Life and Health Insurance Association). [130] Canadian Health Policy Institute, About CHPI. [132] HESA, Evidence, 1st Session, 42nd Parliament, 29 November 2016, 0855 (Ms. Janet Yale, President and Chief Executive Officer, Arthritis Society). [133] HESA, Evidence, 1st Session, 42nd Parliament, 22 September 2016, 0920 (Mr. Jim Keon, President, Canadian Generic Pharmaceutical Association). [135] HESA, Evidence, 1st Session, 42nd Parliament, 2 May 2016, 1550 (Mr. W. Neil Palmer, President and Principal Consultant, PDCI Market Access). [137] HESA, Evidence, 1st Session, 42nd Parliament, 29 November 2016, 0915 (Dr. Doug Coyle, Professor and Interim Director, University of Ottawa, School of Epidemiology, Public Health and Preventative Medicine, As an Individual). [138] Ibid. [139] HESA, Evidence, 1st Session, 42nd Parliament, 20 April 2016, 1705 (Dr. Irfan Dhalla, Vice-President, Evidence and Development Standards, Health Quality Ontario). [140] HESA, Evidence, 1st Session, 42nd Parliament, 9 May 2016, 1540 (Swedlove); and HESA, Evidence, 1st Session, 42nd Parliament, 13 April 2016, 1545 (Hoffman). [141] HESA, Evidence, 1st Session, 42nd Parliament, 20 April 2016, 1705 (Dr. Katherine Boothe, Assistant Professor, Department of Political Science, McMaster University, As an Individual). [144] HESA, Evidence, 1st Session, 42nd Parliament, 2 May 2016, 1550 (Palmer); and HESA, Evidence, 1st Session, 42nd Parliament, 18 April 2016, 1605 (Gagnon). [145] Ibid. [146] HESA, Evidence, 1st Session, 42nd Parliament, 27 September 2016, 0900 (Ms. Mélanie Bourassa Forcier, Professor and Director, Health Law and Policy Programs, Université de Sherbrooke-CIRANO, As an Individual). [147] PBO, Federal Cost of a National Pharmacare Program, p. 29. [148] HESA, Evidence, 1st Session, 42nd Parliament, 23 February 2017, 1140 (Professor Amir Attaran, Professor, Faculty of Law, University of Ottawa). [149] HESA, Evidence, 1st Session, 42nd Parliament, 1 June 2016, 1545 (Mr. Gerry Harrington, Vice-President, Policy and Regulatory Affairs, Consumer Health Products Canada). [150] Ibid. [151] Ibid. [154] HESA, Evidence, 1st Session, 42nd Parliament, 27 September 2016, 0950 (Mr. Ake Blomqvist, Health Policy Scholar, C.D Howe Institute). [155] Ibid. (Forcier). [156] HESA, Evidence, 1st Session, 42nd Parliament, 29 November 2016, 0845 (Dr. Thomas Perry, Chair, Education Working Group, University of British Columbia Therapeutics Initiative). [158] Ibid., 0950. [159] Ibid., 1005. [160] PBO, “Federal Cost of a National Pharmacare Program,” p. 40. [161] Ibid. [162] Ibid., p. 19. [165] HESA, Evidence, 1st Session, 42nd Parliament, 30 May 2016, 1540 (Mr. Matthew Herder, Associate Professor, Faculties of Medicine and Law, Health Law Institute, Dalhousie University). [166] Ibid. [167] Ibid. [168] Ibid., 1550 (McCabe). [169] HESA, Evidence, 1st Session, 42nd Parliament, 20 April 2016, 1545-1620 (Boothe; Dr. Anne Holbrook, Physician/Clinical Pharmacologist, Professor and Director, Division of Clinical Pharmacology & Toxicology, McMaster University, As an Individual). [170] Ibid. [173] HESA, Evidence, 1st Session, 42nd Parliament, 30 May 2016, 1600 (Dr. Robyn Tamblyn, Professor, Department of Medicine and Department of Epidemiology, Biostatistics and Occupational Health, McGill University). [175] HESA, Evidence, 1st Session, 42nd Parliament, 9 May 2016, 1535 (Ms. Anita Huberman, Chief Executive Officer, Surrey Board of Trade). [178] Dr. Anne Holbrook, Director, Division of Clinical Pharmacology & Toxicology, Professor, Department of Medicine, McMaster University, “National Pharmacare: Essential Role of Evidence, Formularies, Expertise” reference document submitted to HESA, April 2016. [179] Ibid. [180] Ibid. [182] HESA, Evidence, 1st Session, 42nd Parliament, 16 May 2016, 1540 (Mr. Perry Eisenschmid, Chief Executive Officer, Canadian Pharmacists Association). [183] HESA, Evidence, 1st Session, 42nd Parliament, 18 April 2016, 1620 (Morgan); and HESA, Evidence, 1st Session, 42nd Parliament, 30 May 2016, 1545 (Boothe). [185] Ibid. [186] Ibid., 1540 (Boothe). [187] Ibid., 1640 (Henry). [188] Pharmaceutical Management Agency (PHARMAC), Introduction to PHARMAC. [190] HESA, Evidence, 1st Session, 42nd Parliament, 14 February 2017, 1110 (Mr. Matthew Brougham, As an Individual). [191] Ibid., 1200 (Ms. Heather Roy, Chair of the Board, Head Office, Medicines New Zealand). [192] Ibid., 1130 (Brougham). [193] Ibid. [194] HESA, Evidence, 1st Session, 42nd Parliament, 2 May 2017, 1200 (Ms. Sofia Wallström, Director General, Dental and Pharmaceutical Benefits Agency). [195] Ibid. [196] Ibid., 1210 (Mr. Aldo Golja, Senior Policy Advisor on Pricing and Reimbursement of Pharmaceuticals, Department of Pharmaceutical Affairs and Medical Technology, Dutch Ministry of Health, Welfare and Sports). [197] Ibid. [198] Ibid., 1315. [199] HESA, Evidence, 1st Session, 42nd Parliament, 18 April 2016 (Morgan, Martin, Dr. Marc-André Gagnon, Associate Professor, School of Public Policy and Administration, Carleton University, As an Individual). [200] Steve G. Morgan, et al., Pharmacare 2020: The Future of Drug Coverage in Canada, 15 July 2015. [202] Ibid. [203] Steve G. Morgan et. al, Pharmacare 2020: The Future of Drug Coverage in Canada, 15 July 2015. [205] HESA, Evidence, 1st Session, 42nd Parliament, 2 May 2016, 1710 (Dr. Graham Sher, Chief Executive Officer, Canadian Blood Services). [207] Steve G. Morgan et. al, “Pharmacare 2020: The Future of Drug Coverage in Canada,” 15 July 2015. [208] Ibid. p. 18. [209] For further details, please see: Steven G. Morgan et. al, “Estimated cost of universal public coverage of prescription drugs in Canada,” Canadian Medical Association Journal, 16 March 2015. [210] Steve G. Morgan et. al, Pharmacare 2020: The Future of Drug Coverage in Canada, 15 July 2015. [211] Ibid. [212] Ibid. [213] HESA, Evidence, 1st Session, 42nd Parliament, 6 June 2016, 1550 (Dr. Gregory Marchildon, Professor and Ontario Research Chair in Health Policy and System Design, Institute of Health Policy, Management and Evaluation, University of Toronto, As an Individual). [214] Gregory P. Marchildon, “Two Options for National Pharmacare,” written submission to HESA, 1st Session, 42nd Parliament, 16 November 2017. [215] Ibid. [216] Ibid. [217] Ibid. [219] Ibid. [221] Gregory P. Marchildon, “Two Options for National Pharmacare,” written submission to HESA, 1st Session, 42nd Parliament, 16 November 2017. [222] Ibid. [224] Ibid. [225] Surrey Board of Trade; CUPE; Canadian Health Coalition; Canadian Labour Congress; Matthew Herder, Associate Professor, Dalhousie University, Canadian Federation of Nurses Unions; Canadian Federation of Medical Students; Canadian Doctors for Medicare; Mr. Peter Macleod, Chair, Citizens’ Reference Panel on Pharmacare; Union des consommateurs; and Registered Nurses Association of Ontario. [227] Ibid. [231] Ibid. [232] HESA, Evidence, 1st Session, 42nd Parliament, 27 September 2016, 0845 (Blomqvist); and HESA, Evidence, 1st Session, 42nd Parliament, 9 May 2016, 1540 (Swedlove). [234] HESA, Evidence, 1st Session, 42nd Parliament, 9 May 2016, 1540 (Swedlove); and Ibid., (Balon). [235] Ibid., (Swedlove); HESA, Evidence, 1st Session, 42nd Parliament, 22 September 2016, 0920 (Keon) and 0935 (Hux); HESA, Evidence, 1st Session, 42nd Parliament, 1 June 2016, 1545 (Forbes). [236] HESA, Evidence, 1st Session, 42nd Parliament, 1 June 2016, 1545 (Forbes) and HESA, Evidence, 1st Session, 42nd Parliament, 22 September 2016, 0935 (Hux). [237] HESA, Minutes of Proceedings, 1st Session, 42nd Parliament, 29 September 2016. [238] RAMQ, Prescription drugs covered. [239] PBO, Federal Cost of a National Pharmacare Program, 28 September 2017. [240] Ibid., pp. 1-3. [241] Ibid., pp. 41-42 (see Table 3-6). [242] Ibid., p. 46. [243] Ibid, p. 42. [244] HESA, Evidence, 1st Session, 42nd Parliament, 17 October 2017, 1530 (Mr. Jean-Denis Fréchette, Parliamentary Budget Officer). [245] PBO, Federal Cost of a National Pharmacare Program, 28 September 2017, p. 32. [246] Ibid., p. 4. [247] HESA, Evidence, 1st Session, 42nd Parliament, 17 October 2017 1535 (Ms. Carleigh Malanik, Financial Analyst, Office of the Parliamentary Budget Officer). [248] Canadian Centre for Policy Alternatives and Canadian Doctors for Medicare, “Cost Savings Resulting from a National Pharmacare Program,” September 2017, p. 4. [249] Ibid., p. 5. [252] Ibid. [253] Government of Canada, Canada Health Act Annual Report, 2015-2016. [254] Ibid. [255] Ibid. [256] HESA, Evidence, 1st Session, 42nd Parliament, 27 September 2016, 0845 (Ms. Colleen Flood, Professor and University Research Chair, Director of the Centre for Health Law, Policy and Ethics, University of Ottawa, As an Individual). [257] Ibid. [258] Ibid., 0900 (Forcier). [259] Ibid. [261] Gregory P. Marchildon, “Two Options for National Pharmacare,” written submission to HESA, 1st Session, 42nd Parliament, 16 November 2017. [262] HESA, Evidence, 1st Session, 42nd Parliament, 6 June 2016, 1635 (Mr. Roy Romanow, Commissioner and former Premier of Saskatchewan, Commissioner on the Future of Health Care in Canada). [264] PBO, Federal Cost of a National Pharmacare Program, 28 September 2017, p. 42. [266] Ibid. [267] Ibid., 1700 (Gagnon). [268] Ibid. [269] For examples, please see: Dr. Marc-André Gagnon, “Chapter 14: Financing the Public Provision of Prescription Drug Coverage in Canada: Comparison and Assessment of the Options,” written submission to HESA, 1st Session, 42nd Parliament, 20 November 2017. [270] Tax Foundation, “Sources of Government Revenue in the OECD, 2016,” July 2016. [271] Ibid. [274] Angus Reid Institute, “Canadian Public Opinion Regarding a National Pharmacare Program,” written submission to HESA, 1st Session, 42nd Parliament, 6 June 2016. [275] Ibid. [277] Ibid., 1605 (Gagnon). [278] Angus Reid Institute, “Canadian Public Opinion Regarding a National Pharmacare Program,” written submission to HESA, 1st Session, 42nd Parliament, 6 June 2016. [281] Ibid. [282] Ibid. [285] The Committee heard or received written submissions from the following patient groups: Canadian Organization for Rare Disorders, Arthritis Society, Canadian Cancer Survivor Network, Mood Disorders Society of Canada, Canadian Diabetes Association, Health Charities Coalition of Canada, and Heart and Stroke Foundation of Canada. [286] HESA, Evidence, 1st Session, 42nd Parliament, 1 December 2016, 0900 (Mr. Michael Ferguson, Auditor General of Canada, Office of the Auditor General of Canada). [287] Ibid., 1000 (Perron). [288] Ibid. [289] Dr. Cara Tannenbaum, Scientific Director, Institute of Gender and Health, Canadian Institutes of Health Research (CIHR), “Policy Brief: Applying Sex and Gender-Based Analysis to Drug Policy in Canada,” written submission to HESA, 1st Session, 42nd Parliament, 20 May 2016. [290] Ibid. [291] Wellesley Institute, “Low Earnings, Unfilled Prescriptions: Employer-Provided Health Benefit Coverage in Canada, July 2015,” p. 8 [292] Dr. Cara Tannenbaum, “Policy Brief: Applying Sex and Gender-Based Analysis to Drug Policy in Canada,” written submission to HESA, 1st Session, 42nd Parliament, 20 May 2016. [293] Canada Gazette, “Regulations Amending the Patented Medicines Regulations,” Vol. 151, No. 48 — December 2, 2017. [294] Ibid. [295] Government of Canada, “Minister of Health Mandate Letter,” 4 October 2017. |
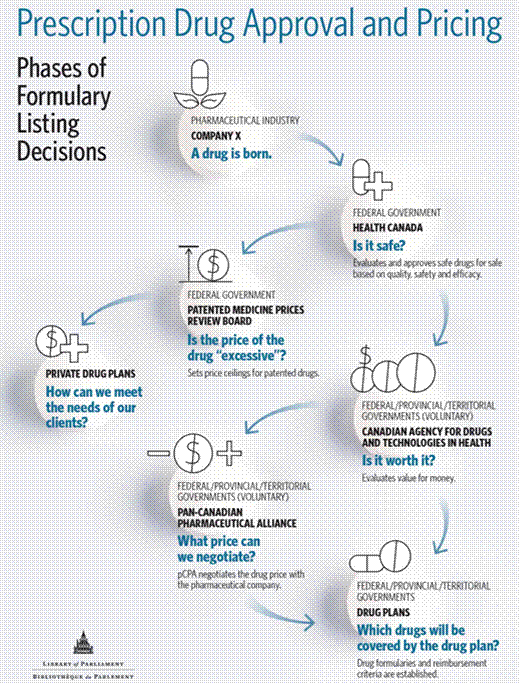
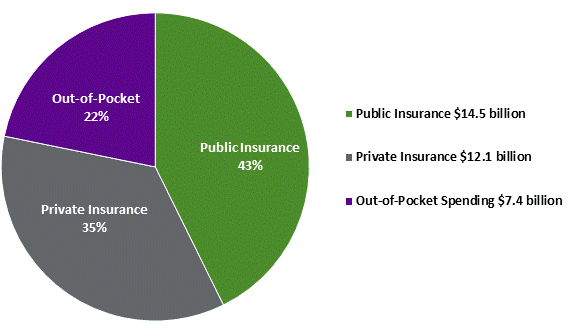
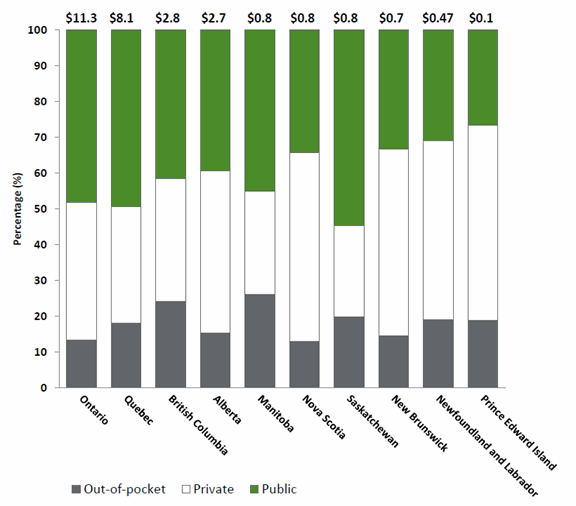
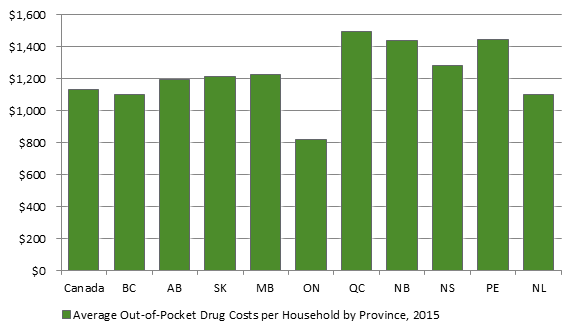
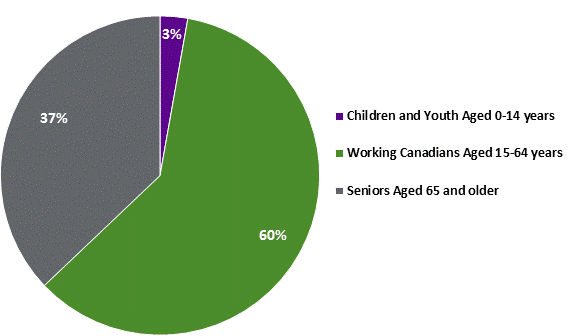
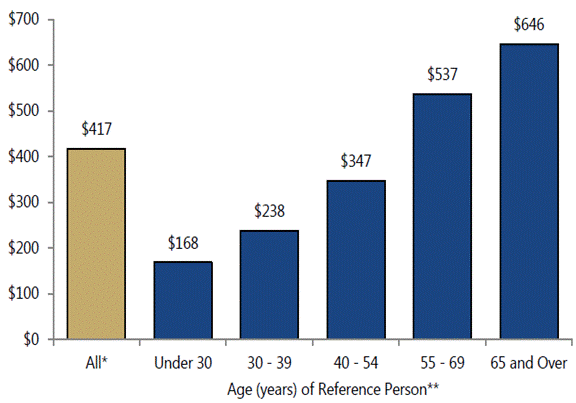
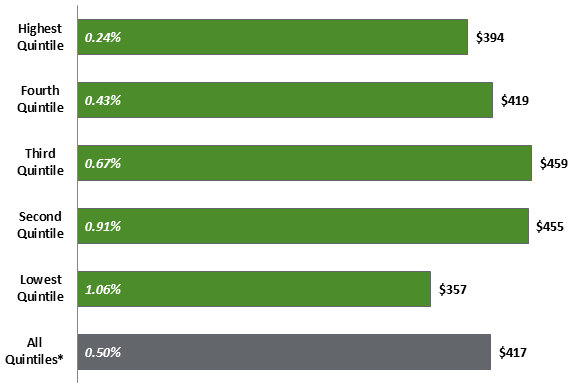
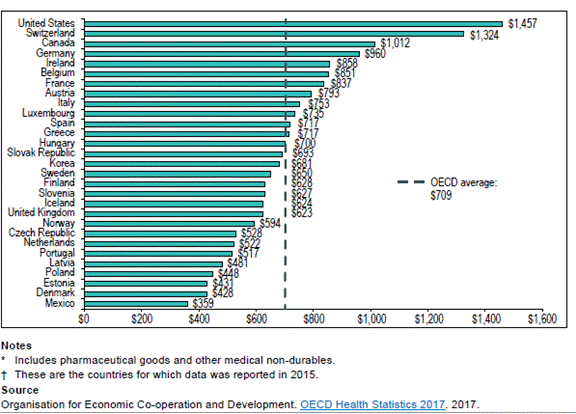
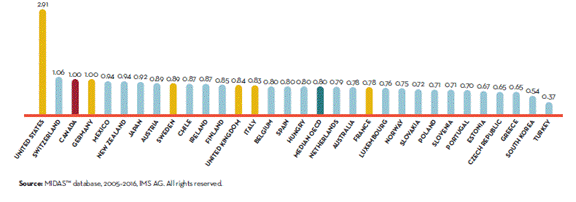
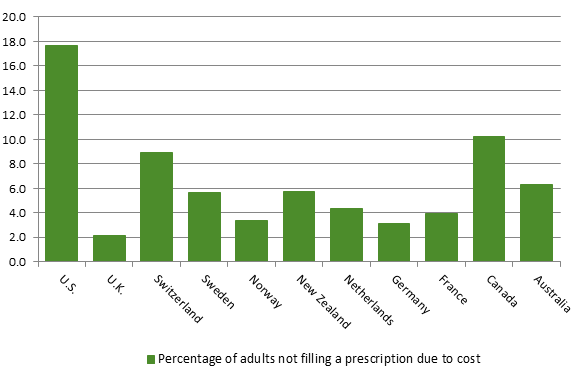

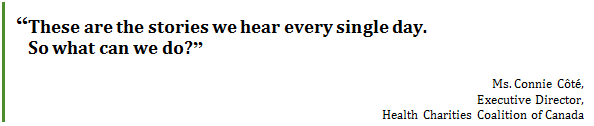

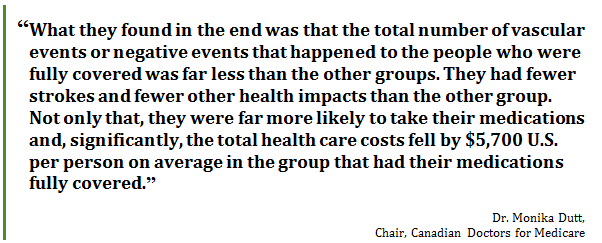



![“If the drug plan [manager] is sitting across the table from manufacturers and saying, ‘It’s our price or you won’t
be subsidized for Canadians across the country,’ that’s a strong bargaining position.”
Dr. Katherine Boothe,
Assistant Professor, Department of Political Science, McMaster University, As an Individual
- Title: Quote - Description: “If the drug plan [manager] is sitting across the table from manufacturers and saying, ‘It’s our price or you won’t
be subsidized for Canadians across the country,’ that’s a strong bargaining position.”
Dr. Katherine Boothe, Assistant Professor, Department of Political Science, McMaster University, As an Individual](/content/Committee/421/HESA/Reports/RP9762464/hesarp14/05-rpt-e_files/pull-8-e.gif)